Qventis unveils their innovative range of hyaluronic acid gels for smoothing fine lines, lifting, and volume augmentation
THE FACIAL HYALURONIC Acid injection market was created almost 20 years ago with the launch of the first hyaluronic acid stabilised gel range (RESTYLANE). The so-called hyaluronic acid dermal filler market has seen less innovation in the passing years as physicians and customers would have like to have seen. From a formulation point of view, the market developed from biphasic products (suspension of cross-linking gel particles — predominantly elastic, in a carrier of non-crosslinked HA phase) to classical monophasic gel formulations (suspension of soft crosslinked HA particles in a carrier of non-crosslinked HA). Basically, all products in the market are stabilised through cross-linking with BDDE. The most important parameter, but not the only one, for the clinical outcome is the cross-linking degree because it determines the biocompatibility profile and lasting effect.
At Qventis we take inspiration from biological systems and make use of science to develop new innovative products. In the extracellular matrix, we find hyaluronic acid chains in a mesh environment with other biomacromolecules — collagen and elastin, where hyaluronic acid is stabilised with pendant groups of proteoglycans. The development strategy while creating RENÉE was to design gel formulations that would minimally affect the rheological profile of native hyaluronic acid. Thanks to the bionic design and an advanced manufacturing process, RENÉE gel formulations are true monophasic, consisting of an homogeneous phase of HA crosslinking gel — free from particles and non-crosslinked HA. The bionic designed RENÉE gels have unique cohesive, elastic, and pseudoplastic behaviour and therefore an outstanding rheological profile.
RENÉE gels are excellent for smoothing fine lines, lifting skin depressions, and restore soft tissue volume augmentation. RENÉE gels are crosslinked with BDDE with a cross-linking degree of 5–6%.

For soft volume augmentation we designed a formulation with 28 mg HA/ml, which is highly resilient and cohesive but still easy to inject. RENÉE gels are easy to inject, to modulate and show good tissue integration in the dermis.
Post-marketing clinical evaluation of safety and effectiveness of RENÉE gels Cristina Esposito MD, Laura Balint MD Study director: Prof. Nicola Zerbinati
The aim of this open label, retrospective, monocentric clinical evaluation was to evaluate safety and effectiveness of the RENÉE range of bionic stabilised hyaluronic acid gels for soft tissue augmentation. The study was done by retrospectively gathering the clinical data of 68 patients treated between October 2017 and January 2018 at the Centro Medico Polispecialistico (CMP) in Pavia, Italy. All treated patients were followed-up for 6 months.
Patient inclusion criteria were:
- for either gender, at least 30 years of age but not more than 70 years of age at enrollment
- congenital or acquired soft tissue deficits of the face with a score of at least 1.5 according to the Modified Fitzpatrick Wrinkle Scale (MFWS)
- a reasonable potential for benefit from correction
- Patient exclusion criteria were:
- being pregnant
- to have had therapy within 3 months prior to the HA injection or have had previous tissue augmentation with permanent implants (e.g., silicone) in the area to be treated
- to be in treatment with anticoagulants or to use aspirin or non-steroidal anti-inflammatory drugs (NSAIDs) within 1 week prior to the treatment
- hypersensitivity to the test product or its ingredients.
Different areas of the face were treated, the Investigator selected the product to use based on the characteristic of the area to treat. The volume injected was 1 ml and no touch-up treatment were performed. The Investigator took pictures of the patients at baseline, immediately after the treatment, 30 days after the treatment and 180 days after the treatment. The pictures were taken with a classic 2D camera (Nikon D90), useful to evaluate the safety of the product, and with a 3D camera (LifeViz Mini) useful to evaluate the difference in facial volume changes.
Results
Clinical efficacy was assessed by two independent Blind Evaluators comparing the pre-treatment pictures with the post-treatment pictures, using two different scales:
- The Modified Fitzpatrick Wrinkle Scale (MFWS)
- The Global Aesthetic Improvement Scale (GAIS).
- The MFWS rating categories were:
- 0 = no wrinkle
- 0.5= very shallow yet visible wrinkle
- 1 = fine wrinkle
- 1.5 = visible wrinkle and slight identation, <1 mm wrinkle depth
- 2 = moderate wrinkle: clear visible wrinkle 1–2 mm wrinkle
- 2.5 prominent and visible wrinkle: more than 2 mm and less than 3 mm wrinkle depth
- 3 = deep wrinkle more than 3 mm wrinkle depth.
- The GAIS rating categories were:
- 1. ‘very much improved’
- 2. ‘much improved’
- 3. ‘improved’
- 4. ‘no change’
- 5. ‘worse’.
The Modified Fitzpatrick
Wrinkle Scale
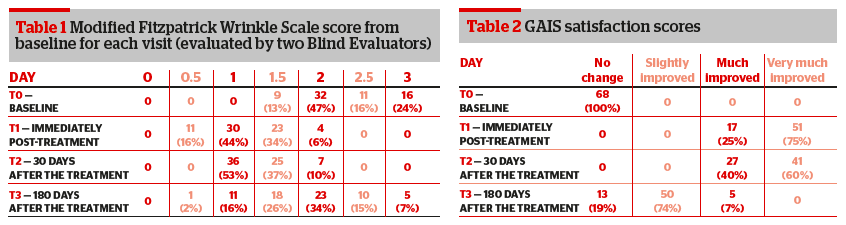
The score at the MFWS before the treatment was:
- 1.5 in 9 patients (13%)
- 2 in 32 patients (42%)
- 2.5 in 11 patients (16%)
- 3 in 16 patients (24%).
Among the 9 patients with a score of 1.5 with the MFWS before the treatment:
- 6 patients scored 0.5 and 3 patients scored 1 immediately after the treatment
- all 9 patients scored 130 days after the treatment
- 3 patients scored 1.5, 5 patients scored 1 and 1 patient scored 0.5 180 days after the treatment.
- Among the 32 patients with score of 2 with the MFWS before the treatment:
- immediately after the treatment 5 patients scored 0.5, 25 patients scored 1 and 2 patients scored 1.5
- 30 days after the treatment 25 patients scored 1 and 7 patients scored 1.5
- 180 days after the treatment 6 patients scored 1, 13 patients scored 1.5, 12 patients scored 2 and 1 patient scored 3.
Among the 11 patients with score of 2.5 with the MFWS before the treatment:
- 2 patients scored 1 and 9 patients scored 1.5 immediately after the treatment
- 2 patients scored 1 and 9 patients scored 1.5 30 days after the treatment
- 2 patients scored 2.5, 7 patients scored 2, and 2 patients scored 1.5 180 days after the treatment.
- Among the 16 patients with score of 3 at the MFWS before the treatment:
- 4 patients scored 2 and 12 patients scored 1.5 immediately after the treatment.
- 7 patients scored 2 and 9 patients scored 1.5 30 days after the treatment.
- 4 patients scored 3, 8 patients scored 2.5 and 4 patients scored 2 180 days after the treatment.
The Global Aesthetic
Improvement Scale
The two Blind Evaluators also rated using the GAIS. 51 patients were judged ‘very much improved’ immediately after the treatment, among these patients:
- 41 patients were still judged ‘very much improved’ and 10 patients ‘much improved’ 30 days after the treatment
- 5 patients were judged ‘much improved’, 40 patients ‘improved’, and 6 patients were judged as ‘no change’ 180 days after the treatment.
- 17 patients were judged as ‘much improved’ by the two Blind Evaluators, among these patients:
- all 17 patients were sill judged as ‘much improved’ 30 days after the treatment,
- 10 patients were judged as improved, and 7 patients were judged as ‘no change’ 180 days after the treatment.
Satisfaction scores
Patient and Investigator satisfaction to the treatment was also evaluated through a self-evaluation questionnaire, that was filled by patients and Investigators 180 days after the treatment, to judge their change in appearance after the treatment. The results were the following:
- 45 patients (67%) were highly satisfied with the treatment
- 21 patients were moderately satisfied with the treatment
- 2 patients had low satisfication with the treatment.
The Investigator was highly satisfied in 50 cases (72%) and moderately satisfied in 18 cases (24%). The Investigator recorded all the side-effects observed during the treatment, immediately after the treatment, and during the follow-up to evaluate the safety of RENÉE gels. No side-effects that required the use of drugs or an outpatient visit were reported by the Investigator.
- 16 patients out of 68 (24%) did not report any discomfort to the filler implant, or adverse effects
- 30 patients (44%) reported slight pain in the implantation of the product
- 22 patients (32%) reported a moderate but still tolerable pain.
Among the 22 patients who reported moderate pain 3 patients (4%) also experienced an oedema, 6 patients (8%) reported oedema and redness that last for few hours. From the analysis of the clinical data we can conclude that RENÉE range of bionic hyaluronic acid gels have a good effectiveness and safety profile. RENÉE hyaluronic acid gels are easy to inject and they are highly appreciated by the patients and doctors. RENÉE range of bionic hyaluronic acid gels offer optimal aesthetic corrective results; no complications, side-effects, local reactions or systemic complications were recorded. In conclusion, patients benefit from the aesthetic outcome, facial rejuvenation effect, well-being and high safety profile when treated with RENÉE range of products.
About Qventis
Qventis is a multi-speciality medical device company, dedicated to develop, manufacture and market novel and reliable medical solutions to boost regeneration. Learning from nature and making use of science, we focus on the design of breakthrough bionic medical solutions based on native or biomimetic ingredients to meet the health needs of an aging population. The company aims to be a provider of tailored regenerative medical solutions for medical aesthetics, joint health, advanced wound care.
Find out more at: renee.com.de