Every field of medicine has its epiphanies — those moments that transform how we think, and what we do as a result. For all of us, the first time we injected a hyaluronic acid filler is one of those moments — compelling evidence that it’s not only our hands, but also the tools in those hands, that profoundly impact the outcomes we can achieve for our patients.
Twenty years of clinical experience with hyaluronic acid, and consolidation of this experience with scientific understanding, have brought many advances. But a true epiphany is different. It’s more than a novel protocol for injection or another way of looking at faces. To paraphrase Peter Drucker, the father of modern management, if we want something new, we have to stop doing something old. The next epiphany for us as injectors is to evolve from using hyaluronic acid for mere space occupation in the tissues, to harnessing its potential as a restorative agent. If we wish this to happen, the tools in our hands are again an essential part of the process.
A hyaluronic acid for skin remodelling
Enter Profhilo, a new hyaluronic acid for skin laxity that is launching in Europe, the UK and elsewhere in the world this year. Profhilo isn’t a filler; it’s the first injectable hyaluronic acid for skin remodelling. In the ten months since Profhilo made its debut in Italy, over 50,000 syringes have been sold in that country alone. So why all the excitement?
The key to Profhilo’s popularity is a unique structure and balance of scientific properties, including a high cohesivity, that allow the product to flow freely through subcutaneous tissue and the dermis, in a way that traditional hyaluronic acid fillers cannot. This makes Profhilo ideal
for anatomically challenging areas that have been difficult to volumize previously, such as the forehead, neck and submalar regions. Profhilo won’t produce lumps and bumps in these or other areas. Even if it’s injected as a bolus or lump, it flows spontaneously within the fat compartments and dermis to fill them evenly within 24 to 48 hours. Profhilo is complementary rather than competitive to other HA fillers. It’s a state-of-the-art tool for skin restoration that we can combine with the fillers we already use to finesse our results.
Profhilo’s unsurpassed level of tissue integration and biocompatibility is due to its unique manufacturing process. Hyaluronic acid fillers are typically stabilized by crosslinking with 1,4-butanediol diglycidyl ether (BDDE), to confer longevity. Otherwise they would be broken down and eliminated within a week or two of injection. In a synergy of scientific and clinical innovation, Profhilo replaces the customary BDDE modification of hyaluronic acid chains with a patented completely chemical-free, thermal technology or process that confers structural and rheologic stability.
BDDE-free technology
While trace amounts of BDDE in hyaluronic acid fillers are considered safe, many companies are engaged in the development of BDDE-free technologies. The concept of injectables that are free of BDDE and other chemicals is very appealing to consumers. Certain regulatory bodies such as the USA FDA have standards for levels of residual BDDE in hyaluronic acid fillers that are lower than those required for approval in other countries. However, the secret to success of BDDE-free hyaluronic acid technologies is that they must offer something that currently available fillers don’t. While Profhilo may hit a chord with consumers because It’s BDDE-free, it’s a hit with injectors because of its performance. It integrates as one into tissue and moves completely naturally with it — even in very mobile areas.
Hybrid cooperative complexes
The science behind Profhilo is fascinating. The technology, developed by Prof. Chiara Schiraldi and Prof. Mario de Rosa at the Second University of Naples, is based on thermally stabile hybrid cooperative complexes of low and high molecular weight hyaluronic acid. It has a high total hyaluronic acid concentration of 32 mg/mL, but a remarkably low extrusion force that makes it easy to inject. Hybrid complexing allows Profhilo to provide greater benefits than each type of hyaluronic acid it contains, without the disadvantages. Profhilo has better active restorative capabilities than low molecular weight hyaluronic acid. However, rather than being pro-inflammatory, Profhilo is actually less inflammatory than even high molecular weight hyaluronic acid, and also has its restructuring capabilities.
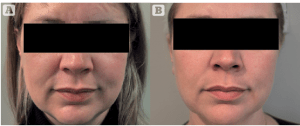
In vitro studies of Profhilo have shown significant up-regulation of collagen and elastin production by both fibroblasts and keratinocytes, with minimal tissue inflammation. Pilot clinical studies performed by Dr. Adele Sparavigna with high-resolution 3D imaging analysis showed a significant volumetric effect at 16 weeks after 2 treatments with an interval of one month of only 1mL Profhilo to the midface on each side. Torsiometry showed significant improvements in skin elasticity and firmness, and patient satisfaction was excellent.
Transforming the way we treat patients
Further scientific and clinical studies are in progress, and yielding intriguing data on this new technology. Profhilo has the potential to take our injectables skills to the next level, and to transform the way we treat our patients. In all areas of the face, neck and décolleté, including those for which we have not previously had the right tools, Profhilo offers a weightless, dynamic volumization and a dramatic improvement in tissue quality.
As the old adage goes, the best way to predict the future is to invent it. Profhilo, the newest and most sophisticated addition to our toolbox of injectables, represents the future of hyaluronic acid, and the future of restorative volumetry.
About the author
Dr. Hema Sundaram is a fellowship-trained, board certified dermatologist and the founding director of her cosmetic dermatology, laser and surgery clinics in Washington, DC. She holds masters and medical degrees from Cambridge University, serves as a Visiting Professor for the American Society for Dermatologic Surgery, and serves on the Scientific Boards of conferences including the International Master Course on Aging Science (IMCAS) and the FACE2FACE congress in Cannes. Dr. Sundaram is an Editorial Advisor to Prime, and a scientific consultant for IBSA Farmaceutici Italia, the manufacturer of Profhilo. She will chair the UK launch symposium for Profhilo, under the aegis of HA-DERMA, at the FACE conference in London this June.
To find out more visit: Email [email protected]; visit us at www.ibsaderma.com; Like us on Facebook: ibsa derma