Daisy Kopera, Zrinka Ivezic-Schoenfeld, Ira G Federspiel, Doris Grablowitz, Benjamin Gehl, and Martin Prinz discuss applications for the hyaluronic acid dermal filler
THE CE MARKED HA DERMAL filler Princess® FILLER is a low viscosity gel consisting of cross-linked HA, designed for injection into the mid-to- deep dermis. The expected lifetime after application is 6–9 months.
Materials and method
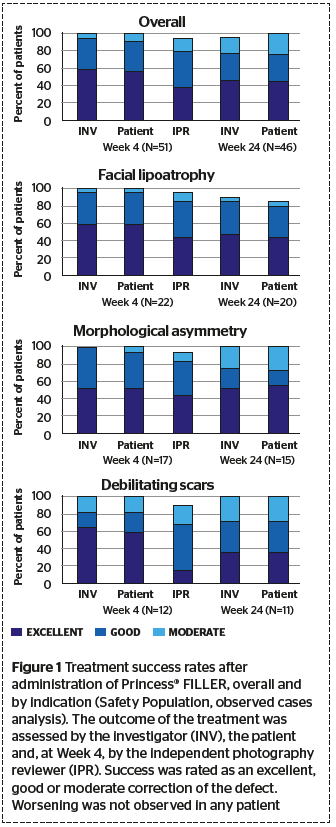
Results and treatment effectiveness
The gained results show that Princess® Filler can restore the pathological deficits in facial or dermal volume. Based on the investigator’s assessment, the treatment was successful in all patients who attended Week 4 visit. In ~95% of the patients, the therapeutic effect lasted for at least 6 months and at this time point the effect of the treatment was graded as excellent or good in around 75% of the patients.
Safety
Eleven (21%) patients stated adverse events. Five (9%) patients reported treatment-related events. Two patients had mild injection site hematoma which appeared 2–3 days after initial or touch-up treatment, respectively, and resolved within 3 weeks. Two additional patients had moderate injection site pain, one of them also reported a headache, and further one experienced headache only. The reported events appeared after touch-up treatment and they resolved within 2–4 days.
Conclusion
- Princess® FILLER was well-tolerated by patients, safe and highly effective
- The overall treatment success rate was 94%–100%
- In most patients, the effect of the treatment was still graded as excellent or good after 6 months.
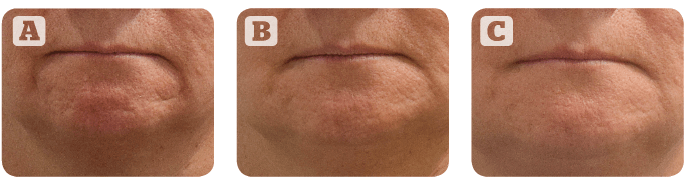
Figure 2 Correction of scars using Princess® Filler. A patient with atrophic scars on the chin, at baseline (A), 4 weeks (B) and 24 weeks after the treatment (C).





