Francesco P. Bernardini, MD, and Morris Hartestein, MD, discuss the oculoplastic perspective of dealing with the complex issues that can arise post-injection

email [email protected]
According to a recent statistic revealed by the American Society of Aesthetic Surgeons, in the past two decades, non-surgical procedures have increased by 186%. However, as injections are increasing in number and areas of the face they can be used, complications are expected to be on the rise. According to a different worldwide survey, the periocular area is the most requested area of treatment by patients and the second most recommended indication by treating physicians.
However, both physicians and patients refrain from a more widespread treatment for fear of visible complications that may alter the aesthetic of such a delicate area, namely lumps, irregularities, long-lasting oedema, and unnatural results.
Classifying periocular complications:
Vascular Complications
Interestingly, despite the close proximity of the peri-orbital area to the eye, the incidence of vascular complications is extremely low. In fact, according to the latest metanalysis of the English literature1 there are no published events of blindness following infra-orbital filler injection. Despite being a low-risk area for vascular accidents, an extensive knowledge of the vascular anatomy is essential.
I often heard well-renowned experts teaching generations of injectors how safe it is to inject deep on the bone with a needle, so I decided to perform a cadaver dissection aimed at evaluating how safe it really was in the infraorbital region, and it turned out that the bone-deep needle injection is anything but safe in this area. First of all, the most commonly mentioned artery of the area, the infraorbital artery, emerges right at the bone level before giving origin to its terminal branches. Here, injectors are faced with various choices:
- Avoid injecting in the ‘danger’ area and stay lateral to the mid-line. This area, however, happens to be crucial, as the home of the deep medial fat compartment (DMFC), and restoring volume to this compartment has been proved key for facial rejuvenation
- Use a 25G cannula, which can be a good choice
- Still inject with a needle, but inject at a more superficial level, by pinching the soft tissue between two fingers and elevating the DMFC from the bone and inject well anterior, i.e. superficial to the IO artery with appropriate fillers. Another common injection site considered safe when approached with a needle on the bone is the orbital rim, and it is frequently used to address the tear trough and the infra-orbital hollow. However, the palpebral artery (PA), a branch of the IO artery, crosses the orbital rim in close contact with the bone before going up to the eyelid, where it contributes to the formation of the peripheral and marginal arcade (Figure 1).
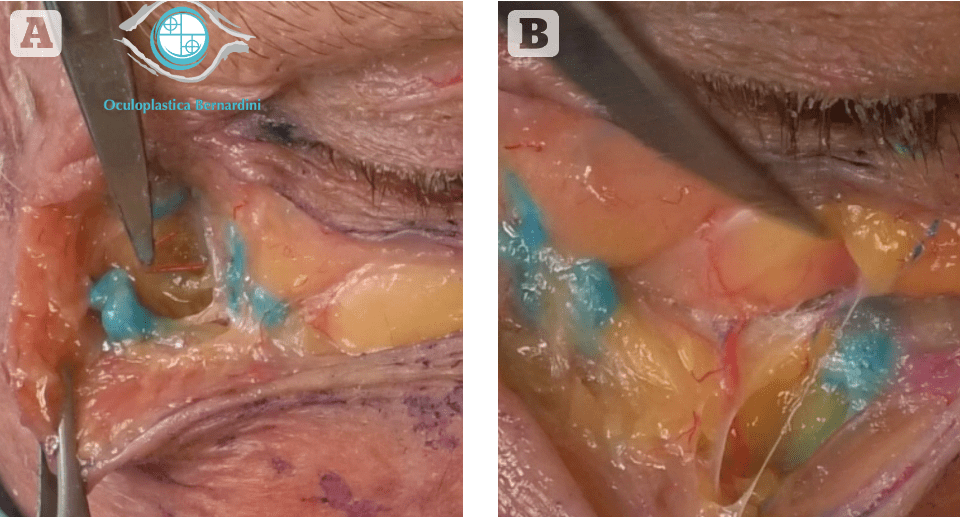
Another interesting point of discordance relative to the safety of bone-deep needle injection, and probably the most frequent point of the face addressed by filler treatment, is represented by the malar eminence. Here, immediately medially and inferior to the malar eminence, lies the emergency of the zygomaticofacial artery (ZFA), a terminal branch of the lacrimal artery, which in turn anastomoses with the transverse facial artery.
Both these arteries, the ZFA and the PA, represent important end-to-end anastomoses between the internal and external carotid circulation, therefore pose a potential risk of blindness if the injection is misplaced. When addressing the infra-orbital area and planning on using a needle on the bone, and maybe feeling safe because you heard somebody famous saying to do so, please keep in mind these vessels and take the due precautions.
Personally, I recommend using a 25G cannula injection always when addressing this area, inject slowly, small quantities per point and never against pain and resistance. Also, when forced by the situation to use a needle, I personally lift the tissue by pinching with two fingers, inject tangentially and purposefully to avoid bone contact. In order to be able to inject ‘more superficial’ in this region, I rely on a dedicated filler.
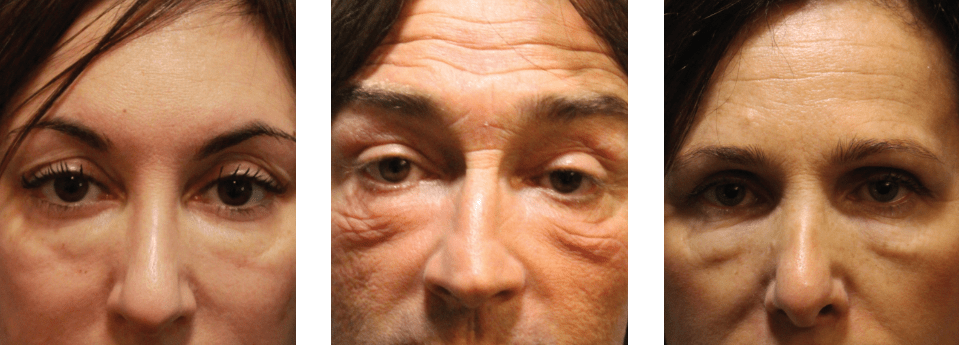
Figure 2 Three examples of chronic delayed-onset lower eyelid oedema, causing a pathognomonic lower eyelid dysmorphism. Always, located at the medial third, bridges the lower eyelid and the cheek, with underlying bluish discoloration, may also alter the skin making it appear wrinkly
Non-vascular complications
Filler indications have expanded to routinely include the periocular region as an attractive non-invasive alternative to lower eyelid blepharoplasty to include not only hollowness but also bags and skin laxity, as we have published2. Recently, the upper eyelid region has become a common target for filler treatments to restore brow volume and projection, ‘elevate’ the tail of the brows, treat the glabellar lines, and correct the sunken upper sulcus. However, HA-based fillers and the eyelids have a complicated relationship, even though biologically compatible, can cause immediate and delayed complications. The most significant and impactful complications include eyelid oedema and DON (delayed onset nodules).
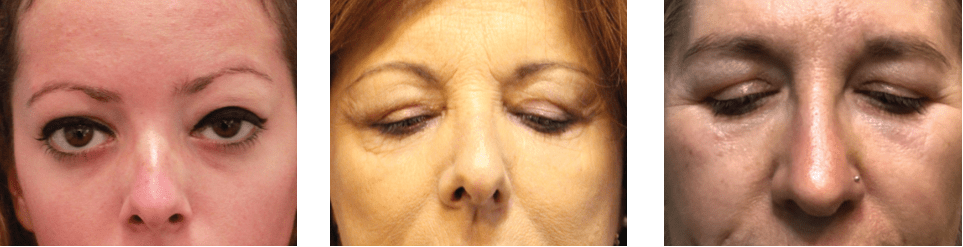
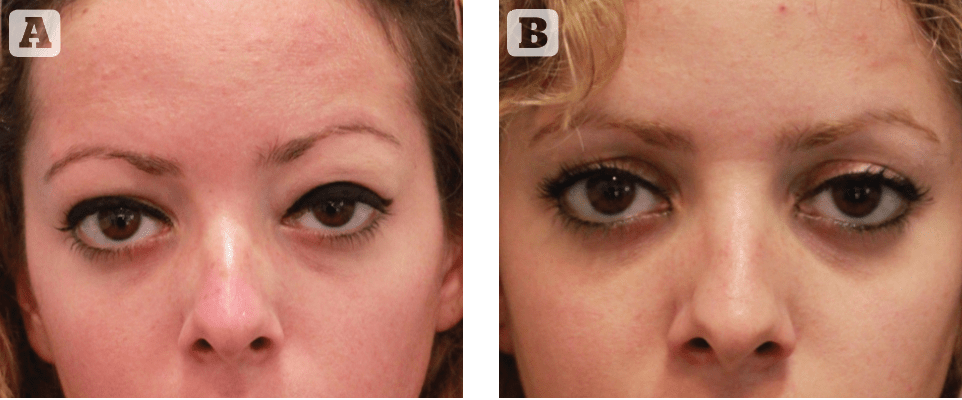
Figure 3 Three examples of upper eyelid chronic oedema, with similar clinical changes as in the lower eyelid, such as localised eyelid swelling, and bluish discoloration. In medicine these changes don’t occur secondarily to any medical condition
Chronic eyelid oedema
Based on the time-lapse from injection and the appearance of the complication, lower eyelid oedema can be differentiated between two different forms: early onset, which occurs soon (within one month) after injection, is believed to be caused by a combination of poor injection technique and/or poor filler selection; normally it tends to resolve in most cases, so a conservative approach is recommended consisting of simple observation or massage. On the contrary, late-onset lower eyelid oedema appears a long time (more than one year) after an uneventful initial HA treatment and tends to progressively worsen with the passing time until it eventually causes an eyelid dysmorphism, defined as an abnormality in the shape or structure of the lower eyelid so peculiar and specific that it can be safely considered, in the filler era, a pathognomonic sign of the presence of commercial HA-based filler (Figure 2).
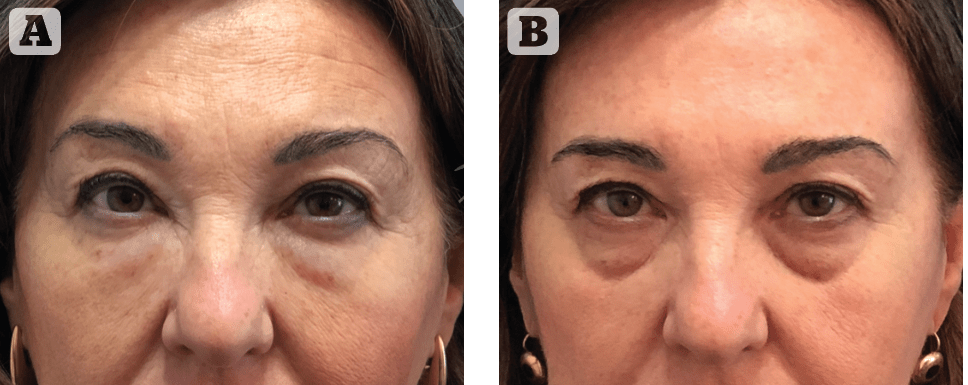
Figure 4 A typical example how the simple management limited to hyaluronidase injection may not be satisfactory for many patients. (A) the patient affected by HA-induced eyelid oedema for three years and unhappy because they look un-naturally too full; (B) the same patient 15 days after dissolution with HYAL, she is still very unhappy because of the presence of her long-forgotten infra-orbital defect
While the term dysmorphic eyelid3 referred originally to the lower eyelid, changes created by HA-induced chronic eyelid oedema, the upper eyelid can also be affected by similar long-standing complications causing a unique dysmorphism4.
The late occurrence of chronic oedema appears to be an HA-related complication limited exclusively to the eyelids only; recognised initially in the lowers and following the trends of expanding indications to filler treatment in the peri-orbital region, it started to play a role in the upper eyelid as well. The clinical manifestations of eyelid oedema are strikingly similar between the upper and the lower eyelids in many aspects. Compared to our recently published paper on late-onset lower eyelid oedema following HA injections3, the time of onset of the upper eyelid oedema is roughly the same, appearing on average 6 to 24 months after the reported treatment and slowly progressing. The appearance of the oedema is accompanied by a bluish discolouration known as the Tyndall effect also in the upper eyelids. Unlike the dysmorphism of the lower eyelid, characterised by one standard clinical presentation of oedema, invariably concerning the medial third, the clinical features of upper eyelid oedema are more variable and reflect the area of injection. In our practices, we have recognised three different patterns of presentation:
- Supero-medial oedema, with localised ‘bulge’, indicating an injection at the level of the glabella
- Centro-lateral brow oedema indicating an injection at the level of the brow and/or temple; and
- Pre-tarsal oedema with eyelid ptosis, also deriving from a glabellar injection. Similar to what occurs in the lower eyelid, where injection at the level of the SOOF of the upper cheek is likely to cause eyelid and malar oedema, we believe that filler injection at the level of the ROOF is the main culprit of the upper eyelid oedema.
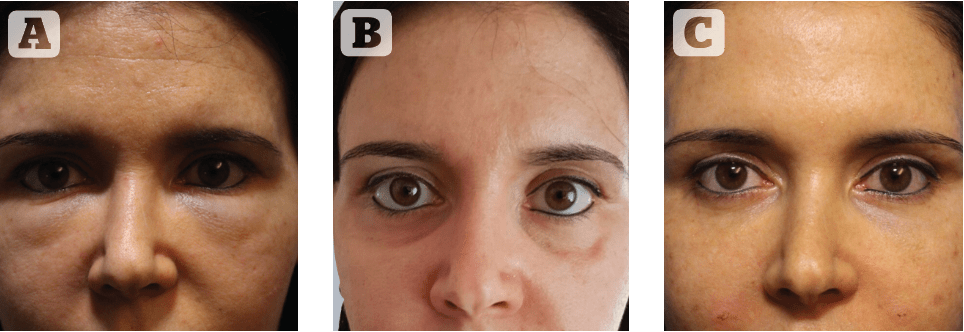
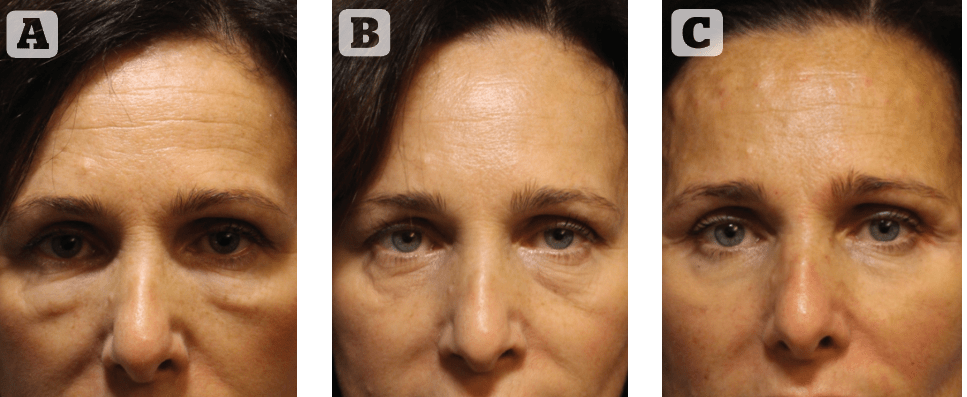
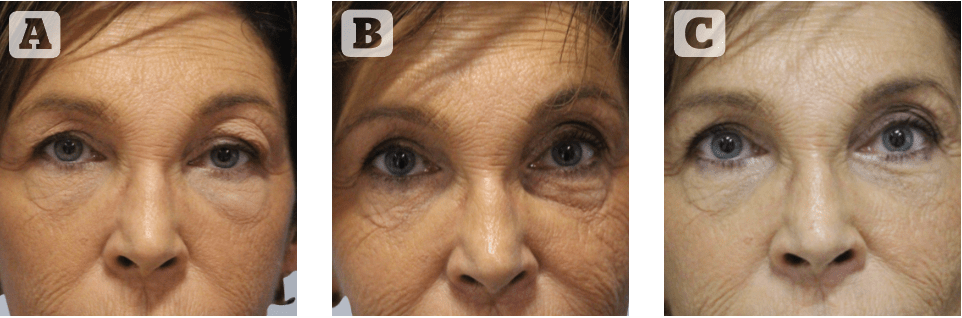
Figure 5 (above) and Figure 6 (below) Two examples of the recommended management for the dysmorphic lower eyelid. (A) both patients showing the pathognomonic signs of HA-induced eyelid oedema; (B) 15 days after HYAL both patients appearing with evidence of hollowing of infra-orbital area, with sunken palpebro-malar hollow (Figure 5B) and deep naso-jugal fold (Figure 6B);
(C) 2 years after secondary filler treatment with satisfactory results
We further suspect that excessive treatment at the level of the glabella can also cause upper eyelid oedema by slowly spilling/diffusing through the supra-trochlear neurovascular bundle/supraorbital notch route to the upper eyelid. This was considered the main mechanism in at least two cases we witnessed recently, where a prominent volumisation was present at the level of the glabella and upper inner canthi, suggestive of this route of extension (Figure 3).
Given the long interval between the injection and the occurrence of the complication, patients often do not relate the problem with the previous HA injection and often deny having been treated. It was also impossible to point the finger towards one or another filler, as patients in most instances don’t recall or didn’t keep track of the utilised filler material, but anecdotally any filler available on the market can be involved.
Regarding the cause of late-onset oedema, the presenting months and even years after an otherwise positive initial injection, some authors support the idea of the complicated infra-orbital anatomy and injection within the malar septum. However, the presence of a similar complication occurring in the upper lid seems to contrast with the idea of a specific lower eyelid or malar anatomy and suggest more general conditions applicable to the entire peri-orbital region. In this sense, I suspect that the major contributing factor resides in the nature of the HA. In fact, it is known that as the HA slowly breaks down, each molecule binds to more water in a way that the same volume can be maintained progressively in a metabolic process known as isovolumetric degradation. It is this progressive water accumulation by the naturally degrading hyaluronan over time that may explain the delayed occurrence of the oedema years after a successful HA treatment. Of course, important questions remain unanswered, such as why does this occur only in the orbicularis oculi laden area? And why do only certain patients experience this reaction and not everybody else?
Late-onset chronic eyelid oedema occurring after uneventful HA treatment is often misdiagnosed as the awareness of this complication and the lag time separating the original injection from the occurrence of the complication may prevent patients and physicians from suspecting the real culprit. This can delay diagnosis and treatment. Further clouding the picture at presentation is the commonly held belief that hyaluronans are self-reabsorbing products that last no longer than 9–12 months approximately, as listed in the manufacturer packaging data. The management option generally accepted included observation, surgery and hyaluronidase. Observation only consists of allowing enough time for the HA to naturally reabsorb and is not effective as time seems only to worsen the condition and I have encountered many cases spanning an 8–10-year duration. Blepharoplasty proposed to physically eliminate eyelid oedema is not indicated in my opinion, as the underlying anatomical defects are concealed by the fluid accumulation and any removal of HA and associated fluid carries the risk of unpredictable results. If surgery is the choice, we suggest that it should be undertaken following the complete dissolution of the HA-induced oedema in order to establish the correct surgical indications. Actually, many cases of cicatricial ectropion that have come to my observation are derived from surgical attempts to address the puffy appearance and the eyelid skin changes induced by long-lasting oedema. The result of this course of action is doomed to catastrophic results as there isn’t a real skin excess and the complete removal of the dispersed filler among the orbicularis muscle is aleatory at best. So HYAL treatment is the best course of action? For what concerns the lower eyelid, it is a little more complicated than this, as dissolving the HA filler restores the baseline condition that initially led the patients to seek treatment (lower eyelid bags, tear troughs, etc.), turning ‘puffy’ unhappy patients into ‘empty’ unhappy patients (Figure 4).
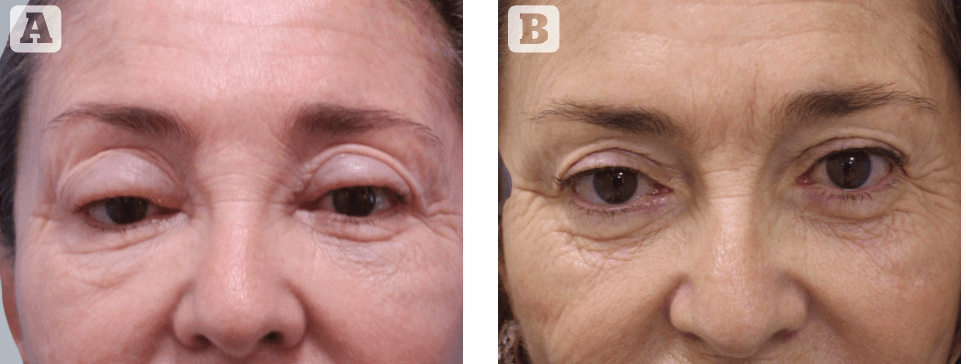
Patients that have not seen their natural eyelids for years, hidden as they were by the oedema, and who experienced a sudden return of their lower eyelid defects after HYAL, cannot be happy with the result unless they have a valid secondary treatment. Figure 7 (A) This case is interesting as the patient presented for an upper blepharoplasty to correct an abnormal appearing left upper eyelid, which appeared abnormally swollen; by looking more closely an abnormal left lower eyelid swelling was also present. (B) 2 weeks after HYAL while the upper eyelid edema appears perfectly corrected, the left infra-orbital region displays a sunken infra-orbital hollow and naso-jugal fold. (C) appearance 15 days after secondary treatment of the infra-orbital area with 2 ml of filler.
This is the reason why for me, it is important to offer a secondary HA filler treatment to be performed two weeks after HYAL, before dissolving the oedema; in our reported series, all patients who opted for secondary filler treatment were satisfied with the final result and didn’t experience any recurrent episode of oedema during the follow-up period (Figures 5–7).
This is very similar to what happens when managing lip filler complications, such as filler migration or over-injection, for example, where filler dissolution is routinely followed by secondary filler treatment. In both cases for eyes and lips, a correct injection technique combined with the choice of adequate filler material is necessary to grant a satisfactory result (Figure 8–10).
Relative to upper lid complications, the treatment with HYAL is more straightforward and resolutive in most cases, without the need for secondary treatment in most circumstances. I found that the diagnosis in these circumstances is what is most challenging.
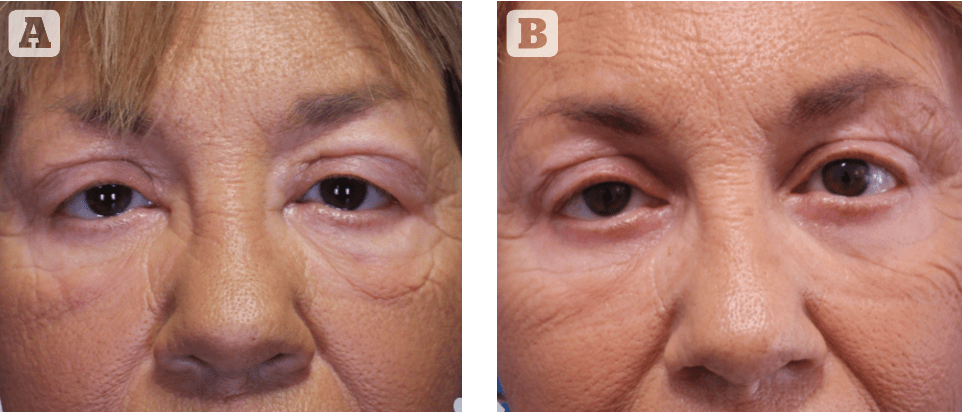
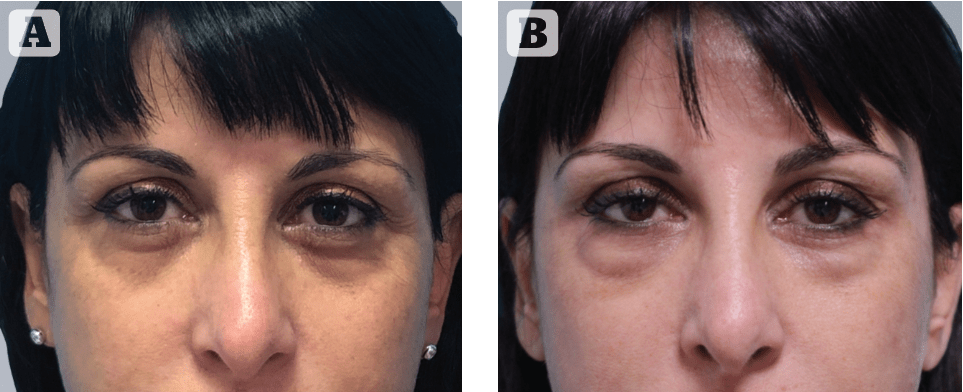
Figure 11 (A) before and (B) 3 months after uneventful filler treatment to the infra-orbital area; the appearance of indurated nodules with signs of inflammation along the infra-orbital rim mimicking orbital fat prolapse, indicating the clinical diagnosis of delayed onset nodules (DON)

Periocular DON (Delayed Onset Nodules)
Finally, the last long-term complication that needs to be mentioned and that we will have to familiarise ourselves with a lot in the near future is represented by the occurrence of delayed onset nodules (DON) in the infra-orbital region. The indurated nodules potential underlying diagnoses include product redistribution, delayed hypersensitivity reaction, biofilm, and granuloma and occur late after the injection. They are considered to occur following a Type 4 delayed hypersensitivity reaction, which is T-cell mediated and can progress to a specific granulomatous reaction to the filler, with some fillers being more prone to their occurrence, according to the literature. They can occur several months after the original injection with or without associated inflammatory reaction (Figure 11).
The occurrence of DON during the Covid vaccinations seems to have increased their occurrence. Their reported treatment consists of administration of intralesional HYAL and may require multiple injections to fully dissolve the nodule.
Declaration of interest None
Figures 1-11 © Dr Bernardini
References
- Beleznay K, Carruthers JD, et al. Update on Avoiding and Treating Blindness From Fillers: A Recent Review of the World Literature. Aesthet Surg J. 2019 May 16;39(6):662-674.
- Casabona G, Bernardini FP, Skippen B, Rosamilia G, Hamade H, Frank K, Freytag DL, Sykes J, Onishi EC, Cotofana S. How to best utilise the line of ligaments and the surface volume coefficient in facial soft tissue filler injections. J Cosmet Dermatol 2020 Feb;19(2):303-311.
- Skippen B, Baldelli I, Hartstein M, Casabona G, Montes JR, Bernardini F. Rehabilitation of the Dysmorphic Lower Eyelid From Hyaluronic Acid Filler: What to Do After a Good Periocular Treatment Goes Bad Aesthet Surg J 2020 Jan 29;40(2):197-205.
- Dubinsky-Pertzov B, Bernardini FP, Or L, Gazit I, Hartstein ME. Late-Onset Upper Eyelid and Brow Edema as a Long-Term Complication of Hyaluronic Acid Filler Injection. Aesthet Surg J. 2021 May 18;41(6)