Giovanni Salti unveils the new portfolio of hyaluronic acid fillers from RELIFE

Definisse™ Filler is a new portfolio of hyaluronic acid (HA) fillers introduced by RELIFE, a company with a solid tradition in pharmaceuticals, being part of the Menarini Group, a leading European pharma group.
HA is the most commonly used material in the production of soft tissue fillers and there are several products available in the market for physicians, that is why we need to dig deeper into its manufacturing process to better understand the technology, the rheology, and therefore the potential clinical benefits of this new HA filler.
Definisse™ Filler
Definisse™ Filler manufacturing is based on a proprietary and exclusive technology named XTR™ (eXcellent Three-dimensional Reticulation). This technology is based on a controlled three step process to achieve a stable and long-lasting filler.
Initially, a controlled fracturing step is aimed to obtain HA fragments of a broad molecular weight starting from a premium quality raw material. In this process, these long chains are fragmented thanks to a controlled thermal process allowing it fragment the original long chain to shorter HA chains. The control of the temperature will enable the manufacturing process to control the length of the HA chains, obtaining two ranges of molecular weight (Medium: 2.5×106 – 3.2×106 Dalton, and High: 3.2×106 – 3.5×106 Dalton).
Successively, the use of a mixture of different lengths of HA chains allows an optimising of the cross-linking process, creating a stable 3D HA matrix. The hyaluronic acid chains of different lengths allows a stable and elastic reticulation. The optimisation of the cross-linking process will enable us to minimise almost to zero the percentage of free HA chains after the cross-linking process. The objectives of obtaining a stable and elastic matrix are to achieve low extrusion force and better-controlled injection, together with effective filling and more durable volumes.
Finally, a highly efficient purification process allows RELIFE to reduce the amount of free BDDE(1,4-butanediol diglycidyl ether). The purification process eliminates almost all the residual BDDE that has not reacted to create inter or intra-chain bridges that reduce the risk of developing reactions after the treatment, resulting in a well-tolerated product with an excellent safety profile.
Definisse™ Filler have been analysed according to standard tests used in literature and have been tested for rheology, cross-linking degree, cohesivity. We also performed a microscopic examination to observe the structural features of the filler scientifically.
Product analysis results
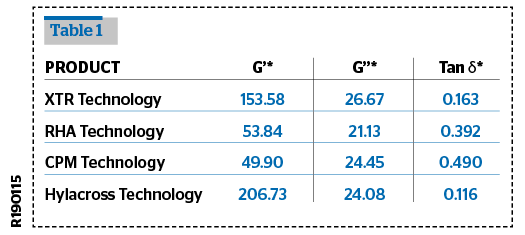
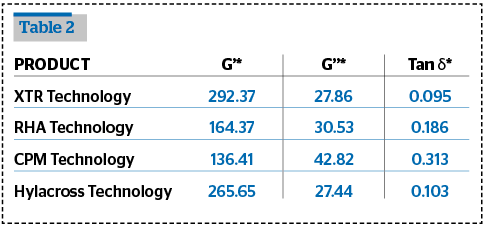
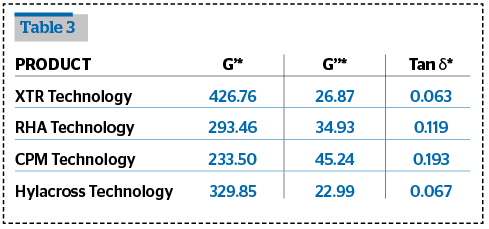
Rheological tests were performed and compared with some of the fillers in the market.
The viscoelastic properties were measured using the Amplitude Sweep tests and Frequency Sweep tests.
The viscoelastic properties have been measured through Amplitude Sweep tests and Frequency Sweep tests.
The rheological analysis was performed in oscillatory flow conditions using a rotational rheometer Rheoplus Anton Paar MCR 101, at a fixed temperature of 30±0.05°C, equipped with a cone-plate geometry CP50-1 or a PP50-P2 sensor.
The determination of reproducibility the viscoelastic properties of HA have been performed using an Anton Paar MCR-101 controlled rate rheometer equipped with a PP50-P2 sensor (50mm parallel plates with serrated surfaces, gap 0,8 mm) and with a Peltier system for temperature control. The results are shown in Tables 1–3.
We examined the cross-linking degree. In particular, the cross-linking ether bridge in comparison with the presence of acetyl groups in the MoD% test (degree of modification). The more cross-link modification is present compared with the acetyl group, the higher the MoD%. The second parameter is the CrR (effective cross-linker ratio) that represents the influence in cross-linked bonds, at both ends, between cross-linkers and HA. The third parameter is a combination of the first two (product’s MoD and CrR), which is indicated as CrD% (degree of cross-linking). All these values show the effectiveness of the cross-linking process.
Cohesivity was identified as an essential characteristic of gel implants. The cohesivity, defined as the capacity of a material not to dissociate, because of the affinity of its molecules for each other, is naturally important during the product distribution into the tissues of the treated area. Cohesivity is considered necessary for the solid and fluid phases of a gel to remain intact, and thus for gel integrity. This scientific study used the five-point visual reference scale (the Gavard-Sundaram Cohesivity Scale) as a reference. Hyaluronic acid filler gel behaviours range from fully dispersed (non-cohesive) with only powder-like gel fragments visible, to fully cohesive with only intact gel strands visible. The video documentations showed visible proof of how the gels were extruded as a single filament from the syringe, in agreement with what expressed by Sundaram et al.
The optical microscopy analyses are performed by LEICA DM 1000 (Leica Microsystems GmbH, Wetzlar, Germany). The filler samples are treated with Toluidine blue-dyed solution 0.1–0.5% w/v and after 2 minutes diluted with deionized water to reach a filler concentration of 50% w/w or 10% w/w. The samples then were imaged at 100x magnification with phase-contrast PH 1, and images are elaborated through LEICA LAS IMAGE ORGANIZER SOFTWARE.
The results from these tests show that Definisse™ Filler have a very high G’ compared to the benchmarked and referenced filler, a degree of crosslinking and a cohesivity in the same ratio of most of the other products, and appears to be more homogeneous on microscopic observation compared to the other fillers.
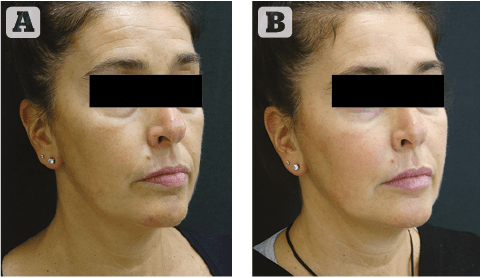
Our clinical experience is limited to 6 months due to the recent introduction of the filler, but we have enough clinical data for a preliminary evaluation.
Based on our clinical experiences, all references of Definisse™ Filler with lidocaine have very good injectability. Especially for the volumiser product (Definisse™ core filler + lidocaine) even when using the micro-cannulas (23G and 25G).
Conclusions
In general, all references of Definisse™ Filler can produce very satisfactory immediate effect. The softer gel (Definisse™ touch filler + lidocaine) is so effective that it requires minimal amounts of product to get a full correction. When using this product, our suggestion is to undercorrect in avoiding excessive deposits. The stronger gels (Definisse™ restore filler + lidocaine and Definisse™ core filler + lidocaine) can produce excellent clinical results even in the long-term supported by their high G’ that allows for excellent projection with small amounts of product. Having a high G’ is the distinct and important feature of Definisse™ Filler since our main strategy of volumisation and lifting is based on deep injection under the SMAS layer, that requires high G’ gels to support the tissues and modulate the muscle activity.
Find out more about Relife at:
www.relifecompany.com
Further Reading
- P. Micheels et al., Effect of Different Crosslinking Technologies on Hyaluronic Acid Behavior: A Visual and Microscopic Study of Seven Hyaluronic Acid Gels, J Drugs Dermatol 2016; 15(5): 600-606
- J. Å. Öhrlund, K. L. M. Edsman, The Myth of the “Biphasic” Hyaluronic Acid Filler, Dermatol Surg 2015; 41:S358–S364
- K. Mondon et al., Influence of the Macro- and/or Microstructure of Cross-Linked Hyaluronic Acid Hydrogels on the Release of Two Model Drugs, J Glycobiol 2016; 5:1.
- Sundaram H et al. Cohesivity of Hyaluronic Acid Fillers: Development and Clinical Implications of a Novel Assay, Pilot Validation with a Five-Point Grading Scale, and Evaluation of Six U.S. Food and Drug Administration-Approved Fillers. Plast Reconstr Surg. 2015 Oct;136(4):678-86
- Salti G1, Rauso R2 Facial Rejuvenation with Fillers: The Dual Plane Technique J Cutan Aesthet Surg. 2015 Jul-Sep;8(3):127-33. doi: 10.4103/0974-2077.167264







