Pronounced localised adiposities may generate variable degrees of psychological embarrassment with potential negative effects on overall quality of life. Fortunately, topical treatments may have a role in reducing unwanted localised adiposities as well as keeping them under control after positive clinical results have been achieved by different medico-surgical procedures. The authors’ have been working on topical formulations aiming at reducing localised adiposities for many years. Their challenge in this study was to try to reduce the amount of time necessary to obtain effective subcutaneous fat remodelling using an innovative vehicle able to optimise trans-epidermal penetration of cosmetic active ingredients. Through morphometric and ultrasonograpic findings, the authors’ could confirm a significant slimming effect induced by active the product after 10 nights of topical application.
Localised adiposities in post-adolescent females are considered important components of an otherwise normal physiological condition and might be interpreted as part of a ‘precautionary body status’ able to guarantee adequate caloric supply in case of pregnancy and lactation. When excessively evident, they can modify female body silhouette beyond normal proportions. These ‘gender-specific fat deposits’ can be observed in approximately 85% of women.
Pronounced localised adiposities may generate variable degrees of psychological embarrassment with potential negative effects on overall quality of life (QOL). Various non-invasive and invasive strategies have been proposed to reduce and control the evolution of excessive amounts of localised fat with variable degrees of success.
Most localised adiposities follow a gynoid type fat distribution and are specifically influenced by oestrogens which stimulate lipogenesis and inhibit lipolysis1–4. Oestrogen-sensitive fat cells have a specific body distribution easily identified on upper and lateral thighs, buttocks, medial knees, lower abdominal regions, and hips.
Normal adipose tissue is physiologically regulated by a highly efficient dynamic equilibrium between fat ‘storage’ (lipogenesis) and fat ‘release’ (lipolysis) in form of FFA and glycerol.
When fat storage is in excess, hypertrophic fat cells can gradually stretch stromal micro-vessels. Should this abnormal micro-anatomical condition persist, a progressive impairment of peri-adipocyte micro-circulation leading to a reduction of oxygen perfusion and derangement of triglyceride metabolism ensues.
Localised adiposities represent still a benign condition and should be considered with appropriate caution when proposing strategies to improve them, avoiding aggressive treatments whenever possible. The multi-factorial origin of this potentially disturbing cosmetic problem should be thoroughly discussed with patients to make them understand the importance of accepting multifaceted, combined treatment strategies involving: prevention, treatment, and long-time maintenance protocols.
Reducing localised adiposities
Topical treatments might have a role in reducing unwanted localised adiposities as well as keeping them under control after positive clinical results have been achieved by different medico-surgical procedures5–8.
The authors have been working on topical formulations aiming at reducing localised adiposities for many years. The authors’ experience allowed them to achieve significant clinical results in terms of reduction of localised fat deposits and their long-term clinical control9–11. Fat reducing processes induced by active topical formulations are relatively long and challenging since their action depend just on cosmetic ingredients able to trigger positive metabolic effects on subcutaneous adipose tissue. The authors’ challenge in this study was to try to reduce the amount of time necessary to obtain effective subcutaneous fat remodelling thanks to an innovative vehicle able to optimise trans-epidermal penetration of cosmetic active ingredients.
The new slimming topical formulation was challenged against placebo to assess its tolerability and clinical effectiveness after one daily application at bed time for a total period of 4 weeks. Clinical results were assessed as early as 10 days after beginning of treatment by clinical and non-invasive instrumental evaluations by a team of dermatologists as well as by self assessment questionnaires completed by all study volunteers.
Materials and methods
Active product ingredients are listed in Table 1.
Study design
The authors’ placebo-controlled double blind randomised study was carried out in two different dermatologic centres: The Laboratory of Cutaneous Bioengineering (Dr M. Mosca, Scientific Director) , Pavia, Italy; and San Gallicano Dermatological Institute, department of dermatology, Rome, Italy. Ninety healthy female volunteers, aged 22–45, with localised fat accumulation on buttocks, thighs, waist, and hips were enrolled in the study. Localised fat accumulations were graded according to a five-point clinical severity scale:
- 0 — no visible subcutaneous fat deposits
- 1 — slight localised skin dimpling
- 2 — localised skin dimpling and depressions
- 3 — localised skin dimpling and depressed striations
- 4 — palpable nodules and depressed striations.
General inclusion criteria were: BMI<30, good general health, and balanced dietary habit and lifestyle.
Exclusion criteria included: allergic diathesis, active inflammatory or neoplastic skin lesions; inconstant body weight; pregnancy, breast feeding, hormonal, and menstrual irregularities; recent hormonal contraceptive regime; early and chronic venous insufficiency; recent (3 months) weight reduction diet; body reshaping procedures (liposuction, radiofrequency, focused ultrasound); and topical slimming treatments.
Study volunteers were asked not to modify their diet and lifestyle habits during the whole duration of the study.
All volunteers were asked to sign a specific informed consent and provide their menstrual cycle details, in order for investigators to properly interpret possible weight variations observed along the study as possibly related to normally occurring hormonal modifications.
During the first, baseline clinical evaluation (T0), each volunteer was blindly assigned a progressive identification number corresponding to the previous treatment randomisation (active vs placebo) secretly set by the formulating laboratory (the active product ingredients can be found listed in Table 1).
Identical plastic tubes containing active and placebo formulations were distributed to each volunteer. Volunteers were instructed to apply test products, on thighs, waist, and hips once a day, preferably at bed time, followed by a 3 minute full hand massage.
Study protocol consisted in four subsequent clinical and non-invasive instrumental assessments:
- Baseline clinical condition (T0)
- On 10th post-treatment day (T10)
- On 15th post-treatment day (T15)
- On 28th post-treatment day (T28).
Each volunteer was evaluated 20 minutes after acclimatisation at standard environmental conditions (18–20°C and 40–60% relative humidity). Body weight was recorded at each clinical evaluation.
Standard anatomical landmarks
Clinical assessments and non-invasive measurements were performed 10–12 hours after topical treatment. Specific anatomical areas identified by a standardised body landmark mapping system (BDMS) was used in order to perfectly compare all clinical data. A flexible measuring tape (cm) was used to measure anatomical circumferences on pre-assigned body landmarks (Figure 1). Measurements were recorded as follows:
- Thigh: midway between hip-bony spine and upper kneecap bony margin (on both sides)
- Hips: at sub-gluteal line
- Waist: at umbilical line.
Ultrasound thickness of all localised subcutaneous adipose layers corresponding to study treatment sites were measured at every clinical evaluation on the same anatomical landmarks (Figure 2).
Ultrasound waves generate back scattering passing through body structures according to their acoustic densities. Their signals can be translated into visual representations according to an A-mode or B-mode scanning resolution. B-mode scanning allows to generate detailed bi-dimensional cross section images of the skin useful to identify its different anatomical macro-components. 10–15 MHz ultrasound sources are usually chosen for skin examination.
Skin thickness can be measured thanks to the time difference between two back scattering signals: (A) skin surface — ultrasound transducer interface; (B) dermis — hypodermis interface. Skin thickness can be calculated multiplying ultrasonic wave transit time between point A and point B by their speed (1540m/s).
Test products were also evaluated by each volunteer in terms of cosmetic characteristics, like fragrance and texture, as well as their clinical efficacy through self assessment questionnaires.
Statistical analysis
No ‘drop out’ was registered among both study groups. Statistical analysis was performed on 89 subjects since data collected from one volunteer belonging to the group treated with the active product showed a protocol violation.
Statistical analysis was therefore carried out on 89 subjects: 44 volunteers belonging to the active product group and 45 to the placebo group.
The following statistical criteria were chosen:
- For normally distributed data, mean value (centrality indicator) and standard deviation (dispersion indicator).
- For data not normally distributed, median value (centrality indicator) and first and third quartiles (lower and upper dispersion indicators)
- Normality was assessed by Shapiro-Wilk test (significance was set at 5%). Variation between paired data, i.e. from T0 raw data and Tn raw data (n = 1–3) was assessed as follows:
- When sample data were normally distributed (Shapiro-Wilk test p-value > 0.05), heteroscedasticity was assessed by Fisher-Snedecor F ratio of variances test
- When normal data were confirmed to be homoscedastic (Fisher-Snedecor test p-value > 0.05), differences in centrality were assessed by Student – Welch paired t test,
- When sample data were not normally distributed (iShapiro-Wilk test p-value < 0.05), heteroscedasticity was assessed by Fligner-Killen test,
- When not normal data were confirmed to be homoscedastic (Fligner-Killen test p value > 0.05), differences in centrality were assessed by paired Wilcoxon-Mann-Whitney test.
The Kruskal Wallis rank sum test was used to assess variations in repeated measures and time profiles.
Results
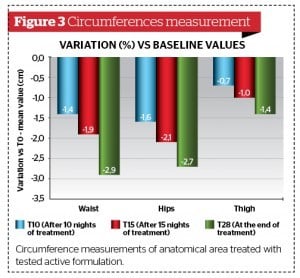
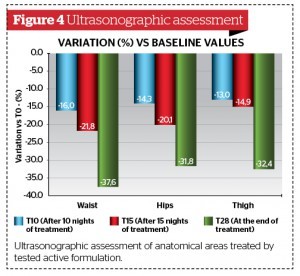
Morphometric (Figure 3) and ultrasonograpic (Figure 4) findings confirmed a significant slimming effect induced by active product already after 10 nights of topical application.
Biometric measurement variations between ‘active’ and the ’placebo’ groups from baseline (T0) showed statistically significant absolute differences between longitudinally collected values using the Wilcoxon test. (p < 0.001).
Volunteers weight was recorded at each visits. Average weight variations were considered compatible with physiological body weight fluctuations independent from the topical treatment performed during the study.
Clinical assessment
Clinical assessment of subcutaneous adiposities located at thigh level was performed using the same five-point severity visual classification scale used as inclusion criteria for each volunteer.
Regular, once a day application of active formulation induced statistically evident clinical improvements at the end of the treatment period (T28) compared to baseline (T0). Clinical evaluation scores improved at least one point in 95.5% of volunteers treated with active formulation (Wilcoxon test p < 0.001).
Subjective assessment
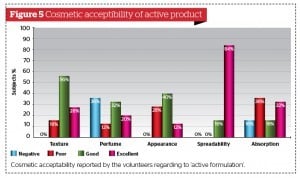
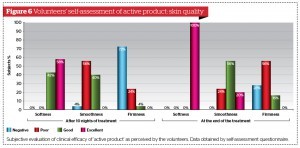
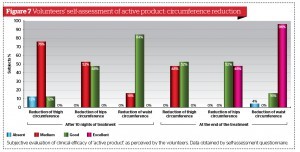
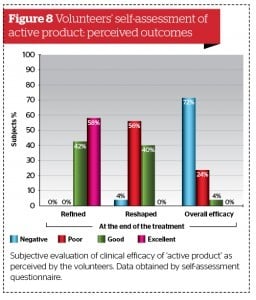
All volunteers were asked to fill out a standardised self-assessment questionnaire focused on personal degree of satisfaction regarding product characteristics (fragrance, easiness of topical distribution, texture, overall appearance, and easiness of trans-epidermal absorption) and skin quality (smoothness, softness, firmness, and tone) perceived 10 and 28 days after once a day continuous application of active formulation and placebo. (Fig. 5 to 8). The majority of volunteers belonging to the active product group confirmed their personal satisfaction on both product characteristics and their skin quality during and after the topical treatment. They were also quite pleased about the circumferences reduction associated with a perceived improvement of their body silhouette on treated areas.
Discussion
The active product tested in this study was formulated to be specifically applied before bedtime in order to work synergistically with the biological effects induced by growth hormone (GH). This hormone is mainly released during the night and is known to activate lipolysis12–18. Other important considerations favouring bedtime topical application of slimming products can be summarised by the advantage on physiological drainage of interstitial fluids given by horizontal body position during sleep; a slightly increased body temperature19; an improved peripheral blood flow20,21; a more active cellular metabolism12–14 ; and an enhanced skin permeability22.
Data from the literature confirmed also that vasoactive ingredients, such as nicotinates contained in the formulation tested in the study, are better absorbed during night time23.
The active formulation tested in the study is a pale green, rich texture, saline emulsion with a good trans-cutaneous absorption .
Its emulsifier system is based on non-ionic ingredients with a high degree of chemical stability even at high concentrations of electrolytes. Key oily phase ingredients are:
- Shea butter, a natural fat extracted from the nut of the African shea tree. This is a rich, complex fatty substance containing many non-saponifiable components such as: oleic acid (40–60%), stearic acid (20–50%), linoleic acid (3–11%), palmitic acid (2–9%), linolenic acid(<1%) and arachidic acid (<1%). Shea butter melts at body temperature and is rapidly absorbed through the skin without leaving a ‘greasy’ feeling
- White clay, rich in minerals such as calcium, iron, potassium and sulphur. It helps to eliminate toxins from the skin, and neutralises free radicals. It also promotes an improved cellular turnover through a micro-exfoliating superficial effect on the epidermis. It is well tolerated even by very sensitive skin types.
Shea butter and white clay work synergistically to improve skin hydration and regeneration.
Functional ingredients were selected with the specific aim of stimulating lipolysis and superficial microcirculation, associated with an efficient draining action of interstitial fluids.
Two seaweeds were selected as lipolytic agents to stimulate triglycerides hydrolysis into free fatty acids, subsequently oxidised by mitochondria:
- Laminaria digitata, belonging to the family of brown algae. Its main constituents are calcium, potassium, iodine and mannitol, as well as fat, protein, carbohydrates, vitamins (E, C, B12, B6, B3, B, A) and other minerals such as Zn, F, Cr, Co, Mn, Na, Fe, P, Mg. Laminaria digitata in its dry form contains 12% water and 15% minerals (chlorines, sulphates and iodines). Iodine in particular, stimulates cell metabolism as well as an increased osmotic exchange leading to elimination of excessive interstitial body fluids. An hydroglyceric extract has been used in the innovative slimming formula
- Gelidium cartilagineum, a red seaweed, rich in rhodystherol. Rhodophyta or red algae contains sterols derivatives able to trigger an effective cellular lipolytic response. An oily extract containing 1.5% active sterols was used in the slimming formula used in the study.
Other functional actives selected to be part of the tested formula were:
- Pure caffeine, a bitter substance found in coffee beans, tea leaves, soft drinks, chocolate, kola nuts. Caffeine is also currently used in specific medical compounds. It has many effects on body metabolism, including a mild stimulation of central nervous system. In the formula, besides its well-known lipolytic activity, caffeine was used to temporary enhance peripheral microcirculation with the purpose of stimulating an effective draining action on interstitial body fluids and making selected tissues more receptive to the active ingredients contained in the formula.
- Pink pepper, derived from characteristic pink berries, sets itself apart from traditional ‘fat burning’ actives owing to its pre-lipolytic action performed on large intracellular lipid droplets. Pink pepper is able to reduce larger intracellular lipid droplet stability, inhibiting Perilipin A synthesis. Its action contributes to keep intracellular fat droplets small making them more suitable to be affected by the action of lipases. Pink pepper can effectively stimulate cutaneous microcirculation without inducing significant vasodilatation. A concentrated oil of pink pepper has been used in the slimming formula.
Selected ingredients with vaso-active properties were chosen to be included in the formula:
- Ethyl nicotinate, a synthetic chemical compound derived from vitamin PP with a erythemogenic effect
- Escin, combined with lecithin to form a special chemical complex, was designed to act on peripheral blood vessels without inducing secondary irritating effects. Escin, extracted from Aesculus hippocastanum seeds, is a triterpenic saponin complex with anti-inflammatory, vaso-constricting and vasoprotective effects. Escin is the main active compound in horse chestnut and is responsible for most of its medicinal properties. Botanical literature indicates escin as a safe and effective remedy for chronic venous insufficiency. A microencapsulated form was used in the slimming formulation.
Ethyl nicotinate and microencapsulated Escin work synergistically to induce temporary peripheral vasodilatation when applied to skin surfaces. This clinically evident effect can successfully decrease tissue toxins through an efficient ‘draining action’ on interstitial fluids; improve tissue oxygenation contributing to enhance cellular metabolism, including lipolysis; and increase positive skin interaction with the other functional ingredients contained in the slimming formula. Escin and ethyl nicotinate, in particular, are responsible for a temporary, mild stinging sensation associated with a short-lasting erythema observed immediately after the application of the slimming formula.
Ethoxydiglycol was selected as a trans-epidermal penetration enhancer to optimise the localised bioavailability of the active ingredients contained in the formula24.
The innovative slimming formula tested in the study relies also on two original strategies:
- A high sea salt content aiming to improve the trans-epidermal draining effect localised on selected skin areas, through a simple osmotic action
- The presence of white clay aiming to form an imperceptibly thin, semi-permeable film on treated skin surfaces to reduce trans epidermal water loss (TEWL) and keep the main active ingredients in close contact with cutaneous layers, enhancing their cosmeceutical action throughout the night.
Conclusions
The clinical and instrumental assessments obtained on the two study groups consisting of standardised circumference and sub-cutaneous fat thickness ultrasound measurements confirmed a localised slimming and body reshaping effect of the active formula over placebo.
No significant slimming effects were observed on the placebo group.
Standardised self-assessment questionnaires completed by the two study groups confirmed the observation of the slimming effect induced by the active product as early as 10 days after continuous once-a-day topical application. Clinical results were shown further improved at the end of the clinical trial (28 days).
The achievement of such an early slimming effect (10 days) on selected body areas was certainly made possible by the effective trans-epidermal penetration enhancer contained in the active formula. This strategy, combined with an innovative combination of cosmetic active ingredients, including pink pepper concentrated oil, was able to induce a consistent localised lipolytic and remodeling effect.
Overall treatment compliance was good in both tested groups throughout the whole duration of the study. The majority of actively treated subjects were satisfied about their skin smoothness, softness and firmness. They were also quite pleased about the circumferential decrease observed on selected body sites, which contributed to generate a cosmetically evident reshaping of body silhouette.
No adverse events or systemic reactions have been reported during the whole duration of the study .
Some volunteers belonging to the actively treated group reported a moderate stinging sensation and a temporary mild degree erythema immediately after the application of the active formula. These symptoms, possibly related to the vaso-active components contained in the formulation, subsided spontaneously without any further treatment and were interpreted ad a sign of ‘efficacy’ by most of volunteers.
This study confirms that well formulated topical cosmetics can be safe and effective in providing clinically evident slimming effects on localised body adiposities.