Wendy Lewis investigates what’s trending in medical aesthetics for the new year and what’s on the horizon
BY ALL ACCOUNTS, THE AESTHETICS market is booming after an exciting and eventful 2018. According to the American Society for Aesthetic Plastic Surgery (ASAPS), 2017 saw a meteoric rise in the top 3 non-surgical categories. The most popular treatment, botulinum toxins, grew 30% since 2012. Hyaluronic acid fillers, ranking second, grew 85% over the same period. Coming in at third place, non-surgical fat reduction expanded at 215% over the past 5 years, notably due to the new players entering this segment and increased availability of clinically validated options for consumers. A key growth area to watch in 2019 will include intimate wellness treatments for women and men.
Additionally, the consumer segments gaining the most traction appear to be millennials (born 1981–1996) and Gen X (born 1965–1980), as well as men. To expand the aesthetics market requires two main drivers; first, by attracting more patients to get on board the treatment train. Secondly, to encourage current aesthetic patients to increase the treatments they are already undergoing by way of expanded applications, by having the same treatments at more frequent intervals, and/or adding new treatments to their regimen.
Injectable trends
A newcomer among FDA cleared neurotoxins in the form of prabotulinumtoxin A, conditionally named Jeuveau™ from Evolus™, is awaiting FDA approval in 2019, with more on the way. Revance® Therapeutics’ longer lasting DaxibotulinumtoxinA (RT002) is also on the path to a US launch potentially for 2020. Allergan® picked up the fast acting, shorter duration toxin BoNT/E that may be positioned for first time users in the near future.
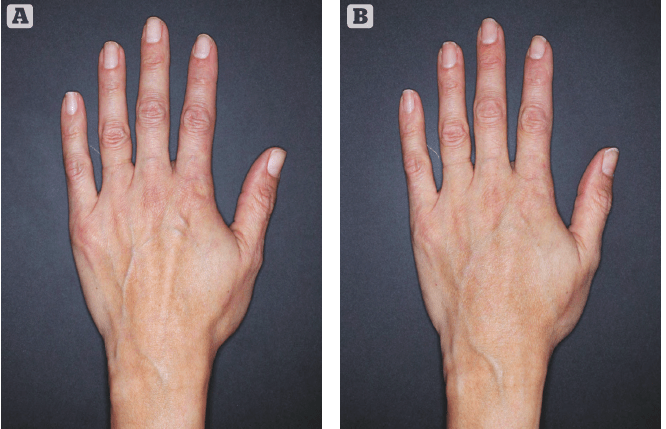
Figure 1 Treated with 3mL of Restylane Lyft in the left hand. (A) Before. (B) 4 weeks after treatment
Allergan and Merz® Aesthetics are both going after new audiences to grow the injectable market. The younger toxin market is up for grabs and campaigns targeting patients in their 20s–30s have the potential to expand the number of users considerably. Allergan launched a bold ‘LIFT IT, SMOOTH IT, PLUMP IT, JUVEDERM® IT’ campaign to reach this emerging group of injectable customers. Merz recently launched a dramatic Xeomin® campaign ‘Later Haters,’ targeting a slightly different audience of women they call ‘The Reclaimers,’ who have spent the last several years building a career or raising a family and are ready to refocus on taking care of themselves.
Beauty-savvy consumers are starting earlier with micro or mini treatments of 5 units of toxin strategically placed to prevent the formation of dynamic wrinkles, as in the glabellar and crow’s feet.
Micro treatments are on the rise as the perception among consumers is that a series of small tweaks can add up to big improvements down the road. ‘Micro Botox,’ also referred to as ‘Baby Botox’ in some circles, is leading this trend as being used to help decrease oil production and pore size and improve skin tone. This concept of ‘prejuvenation’ or early intervention ranks high among this emerging group of clients entering the aesthetic treatment market.
More practitioners are using Kybella® (Allergan) or Belkyra® outside of the USA, in an off-label manner to treat fat deposits on non-facial areas, including upper arms, axillary, back, abdomen, flanks, thighs, knees, calves, and ankles, as well as for touch-ups, post-invasive body contouring, and in combination with non-surgical fat reduction therapies and skin tightening.
The most recent hyaluronic acid filler launched in the US market in January 2018 was Revanesse® Versa™ (Prolennium Medical Technologies). Another first was the FDA approval of Restylane® Lyft (Galderma) for the correction of age-related volume loss in the back of the hands. We can expect new regulatory approvals of expanded indications, including more off-face uses for currently marketed fillers in the US in the near future.

Emsculpt® by BTL Aesthetics®
Platelet rich plasma (PRP) seems to be steadily increasing in patient and practitioner acceptance as well as its applications. It is being used along with many other procedures including microneedling as a stand-alone treatment for skin rejuvenation and hair regrowth, as well as in conjunction with fat grafting and post-laser resurfacing.
Microneedling combined with radiofrequency energy has gone mainstream, and is now widely used for addressing skin rejuvenation, acne scarring, stretchmarks, and cellulite.
Energy based devices
In this segment, non-surgical fat reduction and body contouring seem poised to be the most dynamic segments moving forward. For starters, they cast the widest net among both genders and age groups. These are also considered the most robust categories in terms of the range of treatments available, new technologies emerging, novel products, and a wide range of fees that make them suitable for many aesthetic patients, even those just entering the market.

Renuvion® Cosmetic Technology, from Apyx Medical
BTL Aesthetics® rocked the aesthetics market in 2018 with the introduction of Emsculpt® that uses high-intensity focused electromagnetic (HIFEM) energy to induce muscle contractions and destroy fat cells. Stay tuned for more innovations to stimulate muscle contractions utilizing t ranscutaneous electrical nerve stimulation (TENS), electric muscle stimulation (EMS), and combinations of low voltage currents with fat reduction. Cutera® introduced their 3rd generation of fat sculpting technology, truSculpt® iD, that uses monopolar RF in a hands-free model that can treat a full abdomen in 15 minutes.
Plasma regeneration is making a comeback. For example, Renuvion® Cosmetic Technology, from Apyx Medical (Formerly Bovie Medical) and Nitrogen Plasma Skin Regeneration with NeoGen™ from Energist® are generating interest among plastic surgeons to aesthetic doctors for the dramatic improvements that can be achieved in a single treatment. Results are particularly notable in the periorbital and perioral areas, which are historically difficult to treat.
A new generation of non-energy-based treatments is also emerging. A minimally invasive micro-coring skin removal technology from Cytrellis® Biosystems, currently under investigation, shows promise for removing sagging skin. Another newcomer is the Recros Medica™ rotational fractional resection (RFR) called Nuvellus™ that combines fractional skin resection and lipectomy.
Intimate wellness
truSculpt® iD fat sculpting treatment by Cutera
Among the evolving treatments in this growing category, low-intensity extracorporeal shockwave therapy (Li- ESWT), commonly known as shockwave therapy, is now being used off-label to treat erectile dysfunction. Among the other devices targeting men’s erectile dysfunction are Zimmer’s ZWave® radial shockwave unit, and eVIVE™ from Eclipse, which uses acoustic sound wave therapy. These treatments are finding their way into more aesthetic practices offering intimate wellness solutions.
Although the vaginal rejuvenation and women’s health category attracted the attention of the US FDA in 2018 and faced scrutiny, the expanding range of energy-based treatments remain in high demand from women of all age groups, from post-childbirth to perimenopausal. Bioidentical hormone therapy with oestradiol and progesterone are often used in combination with energy-based treatments. PRP is also now frequently used for intimate enhancement procedures, such as the ‘O-Shot®’ or Orgasm Shot, and ‘P-Shot®’ as in Priapus Shot. Home care vaginal wellness systems, such as vFitPLUS™ (Joylux Inc) or VSculptPRO™ outside of the US, are also gaining interest from women in search of solutions.

vFITPlus™ by Joylux Inc
2019 promises to be an exciting year for the aesthetics industry. Watch this space.







