Dr Joan Vandeputte reviews the current and future role of fillers in aesthetic medicine, including indications, potential problems and areas for further research

Joan VANDEPUTTE
Vandeputte is a plastic surgeon at the Oudenaarde General Hospital (A.Z. Oudenaarde) in Oudenaarde, Belgium and in private practice at Meerspoort 31, 9700 Oudenaarde, Belgium
email [email protected]
Generations of plastic surgeons have considered the removal of obvious bulges and the treatment of sagging as principal paradigms in facial rejuvenation. The strategies came down to excision and shifting of soft tissue volumes. Over the last decades, the treatment of volume deficiencies by fat injection at various levels of depth1 has successfully expanded the armamentarium. In parallel, fascinating techniques were developed by non-surgical and surgical colleagues to compensate volume loss by injection, at various levels, of fillers, some of which are biostimulatory2,3.
Besides the hollow eyes and tight-pulled faces resulting from surgery4, chipmunk cheeks, extravagantly pouting lips and inflated faces have now also appeared in our communities, on occasion also in professionals in the field of medical aesthetics.
While the numbers of indications are expanding along with the number of therapeutic options, achieving a predictable outcome, avoiding excesses and accurate risk management remains a challenge.
Materials and methods
Representative clinical photography from the author’s practice and of his cadaver dissections were compared to data from the literature, reflecting on the evolving place for volumising injections in facial aesthetics and points of interest for studies and clinical training. The author chiefly used the following fillers.
The Belotero* portfolio consists of non-particulate hyaluronic acid (HA) gels. The Restylane** portfolio comprises particulate HA gels. All are derived from bacterial HA and cross-linked with 1,4-butanediol diglycidyl ether (BDDE). The cellular tissue response is low. After hydrodynamic and enzymatic break-down locally, endogenous HA is metabolised in the lymph nodes, the liver and the kidneys while a small portion is excreted in the urine5. HA gels probably follow the same pathway in a delayed manner. Clinical and MRI studies show HA gels to stay in situ for years6,7 after deep injection.
The Algeness*** portfolio are particulate gels of agarose from Gelidium red algae. The purified agarose is not chemically modified. The local cellular response is high, as the filler is resorbed in situ by macrophages without capsule formation. Little has been published on the longevity of results. The author observed clinical effects that lasted well over a year.
Radiesse**** is a suspension of calcium hydroxylapatite microspheres in a carboxymethylcellulose gel. The gel is resorbed in a few months, while the biostimulatory effect of the resorbing spheres, inducing microcapsules, last for over a year3.
Sculptra***** is a powder of a poly-L-lactic acid (PLLA) polymer, suspended in water for injection before use. It has a strong biostimulatory effect by encapsulation and fibroplasia. As it is slowly resorbed, it is categorised as a semi-permanent filler3.
Results and discussion
Applications for volumising fillers
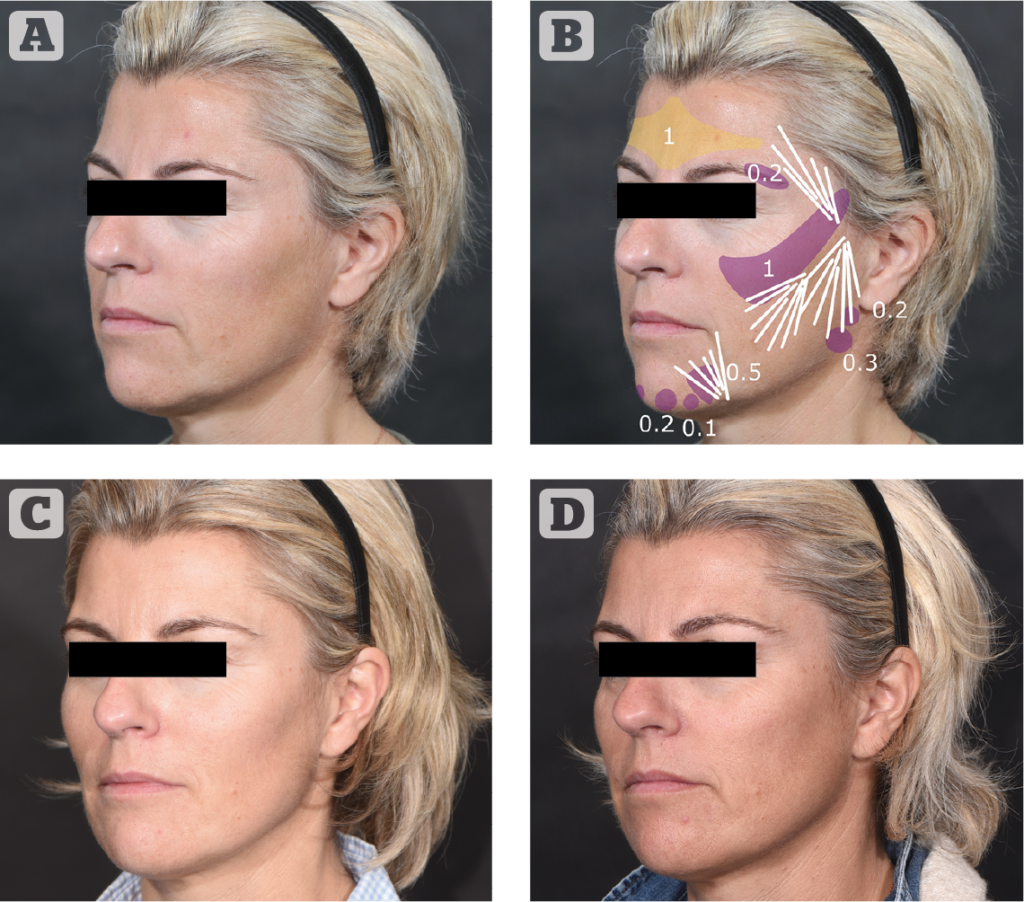
Volumising fillers can be used to compensate for acquired or constitutional bone or soft tissue volume deficiency (Figure 1). Increasing projection (e.g. eyebrows, cheekbone, mandibular angle, chin) may require more focal deposition of filler, while diffuse filling of excessive concavity and smoothing of sharp surface boundaries requires even deposition over larger surfaces.
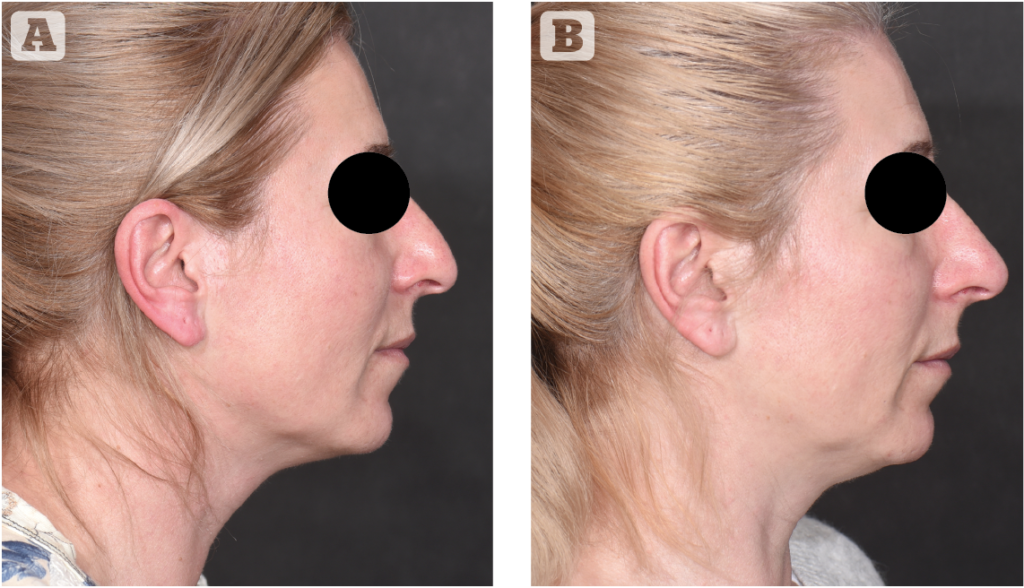
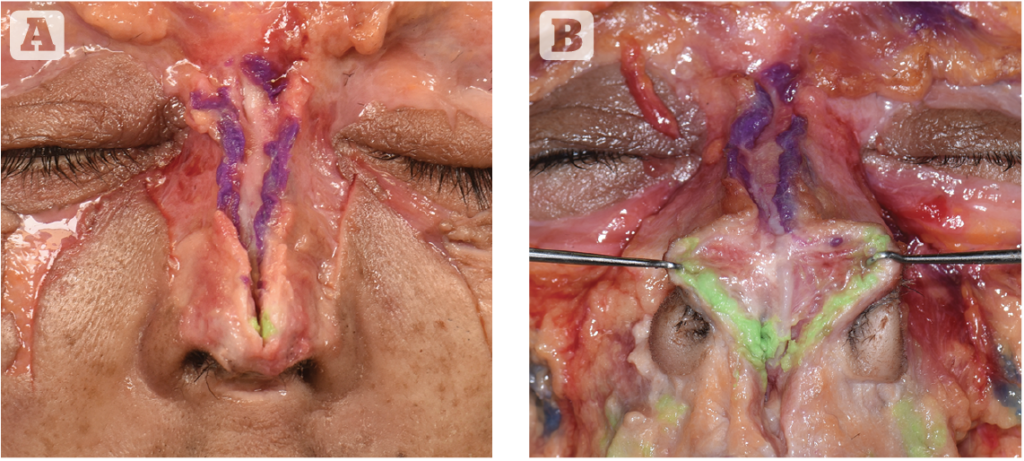
Volumising fillers can also provide support to overlying structures. The treatment of a slightly drooping nasal tip (Figures 2–3) is a typical example.
Safety
The most frequently reported severe complication of fat or filler injection is necrosis8,9, followed by a worrying number of cases of blindness8–11. The highest incidence among fillers is reported for HA7,8, but this is also by far the largest group by number of injections12. Preventive measures and recommendations for treatment were listed extensively by Belaznay et al11. Infections of injected areas and granulomas8,13, if less catastrophic, are still serious although less rare complications.
HA’s have the distinct advantage that they can be removed fast from the interstitium by hyaluronidase injection. To some extent, intra-arterial HA can be dissolved by hyaluronidase injection in and even around the vessel9.
Where is the filler
going during injection?
Comparison of MRI to dissections in 40 cadaver heads, after injection with MRI contrast medium mixed with food dye, by Cotofana et al.,14 showed no displacement of filler outside the intended area of injection. However, work by Pilsl et al.,15 indicates otherwise. They injected the premaxillary (Ristow’s) space in 15 cadaver heads with calcium hydroxyapatite in carboxymethylcellulose versus a less viscous dye designed for intra-arterial injections for anatomical dissections. The dye spread into the malar fat pad (the deep medial fat pad). They concluded that the premaxillary space is semi-open. Pavicic et al.,16 demonstrated, in a cadaver study, that needle calibre and, even more, angle of injection, influence filler spread along the needle trajectory. Figure 4 confirms how a filler, injected perpendicular into the temporal muscle, can spread to a superficial level.
Cannulas can distend septae which, on retraction, shift the injected bolus to a more proximal position along the trajectory17.
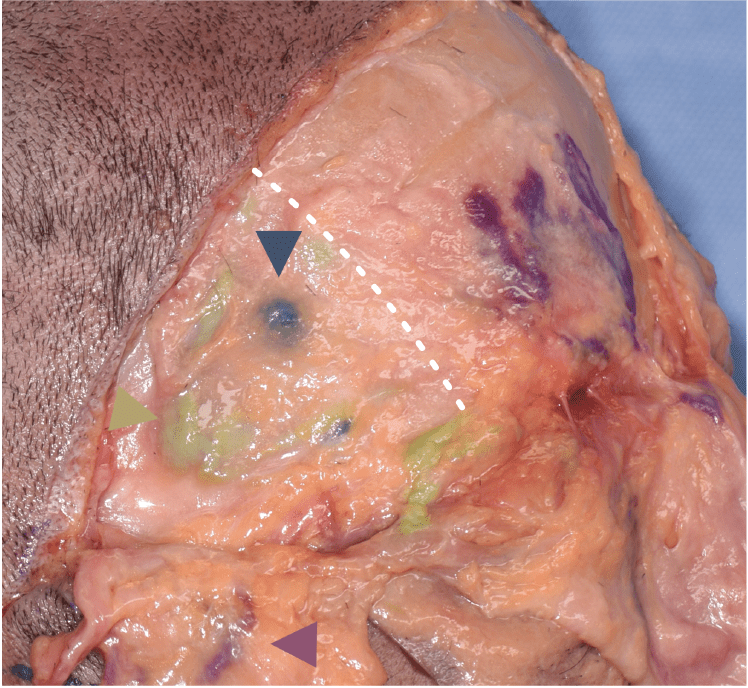
Figure 4 Cadaver dissection of the right temple after injection of dyed agarose 3.5% (Algeness DF) (blue) in the temporal muscle with a sharp 27G needle after making bone contact, 2.5% (green) deep subcutaneous (under the superficial fascia) with a cannula 25 and 1.5% (purple) subdermal with a cannula 25. The dotted line indicates the position of the temporal crest. The superficial subcutis containing the 1.5% (purple) has been dissected off the superficial fascia. Not only do we see the green 2.5% filler shining through the superficial fascia. The blue 3.5 % filler injected deep in the temporal muscle has travelled along the trajectory and some is in a deep subcutaneous position.
We can therefore not take for granted that all filler is deposited exactly where we intend, nor that it cannot spread instantly in an undesirable direction.
Where is the filler
going after the injection?
The ideal volumising filler would not migrate under external pressure and resume its original shape when the pressure is released. While its elasticity can be measured in vitro, we do not know yet if filler rheology has a measurable impact on the elastic behaviour of injected tissues.
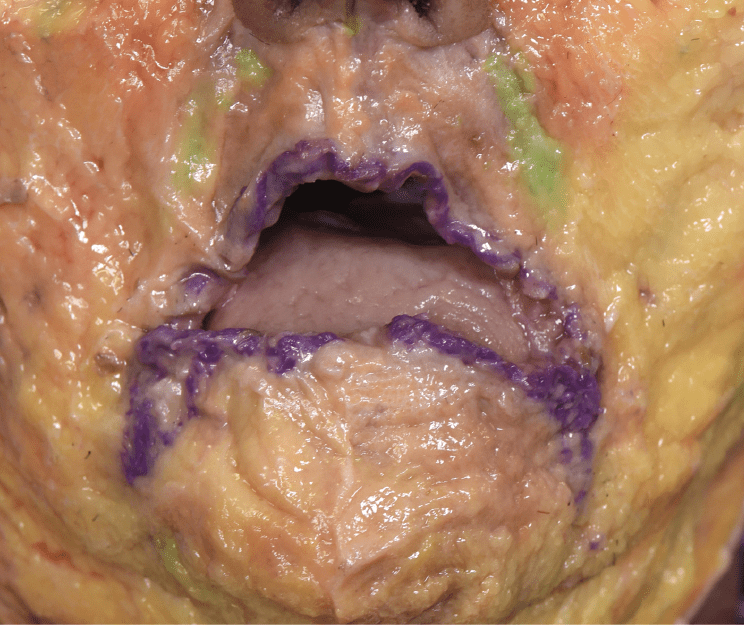
Although remodelling of the injected zone by finger pressure is common practice and described in the literature6,18, we need more research to find out if external compression can effectively do more than change the shape of the deposit without displacing it and expulsing a reactive oedema. Cadaver dissections after injections with dyed agarose gel showed no apparent effect of firm remodelling (Figure 5).
Lumps and swelling
Lumps, besides cases of infection or granuloma, can originate from the irregular deposition of filler, water absorption by the filler, reactive oedema or tissue response with cellular components, with or without capsule formation.
A contour deformity can only be focal. A good example is a small, unintended, subdermal deposit occurring during deep injection on the orbital margin. The thin skin of the temple does not easily hide irregularities. Lambros advocated diluting hyaluronic acid dermal fillers with saline for subcutaneous, temporal injection because of the high probability of lumps in this area19. A deformity can be local, including the entire filler deposit and the immediate vicinity, such as sometimes happens after HA injection over the orbital margin. Third, contour deformities can also be locoregional, such as oedema in the wider surroundings of the injectable, with a puffy look.
Precision and predictability
To avoid clogging of the needle by particles, poly-L-lactic acid suspensions need to be prepared well in advance and at sufficient dilution3. The product has to be spread out as evenly as possible throughout the target area to avoid the formation of visible and palpable lumps, with a higher dilution for subcutaneous than for deep treatment. The low viscosity and the imperative diffuse spread make it less suitable for circumscript corrections. The biostimulatory effect is strong, but the result depends on tissue response, which takes time and, like any biological phenomenon, is variable.
The viscosity of calcium hydroxylapatite and the more subtle tissue response makes it more suitable for circumscript corrections, while diffuse infiltration can be performed just as well. The result at mid and long term is not uncommonly appreciated more after a repeat treatment. Immediate reactive oedema can be pronounced, but mid- or long-term oedema is very rare.
It is not recommended to inject biostimulants in the glabella, on the orbital margin, in the lips or in the nose because of the risk of direct visibility of hard lump formation and for safety reasons.
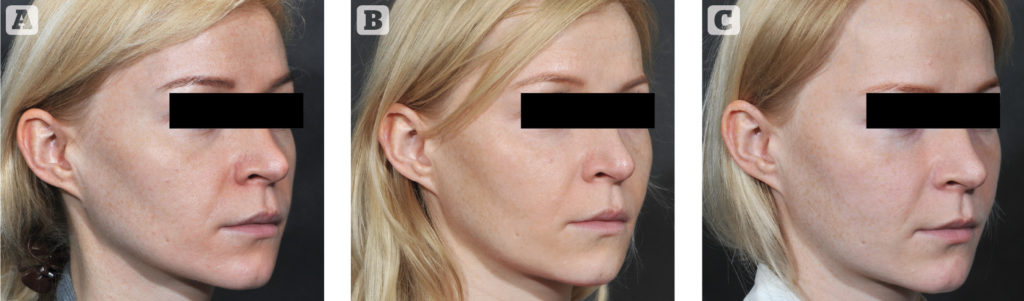
The vast array of HA gels offer great versatility in subcutaneous to deep injection, to increase projection or for diffuse filling. Reactive oedema immediately after injection is limited but mid- and long-term oedema, especially in the periorbital area and the lips, can be an issue. Soft irregularities may occur, especially in areas with thin skin19. In some individuals there is some loss of projection over time and some volume increase extending beyond the injected areas (Figure 6).
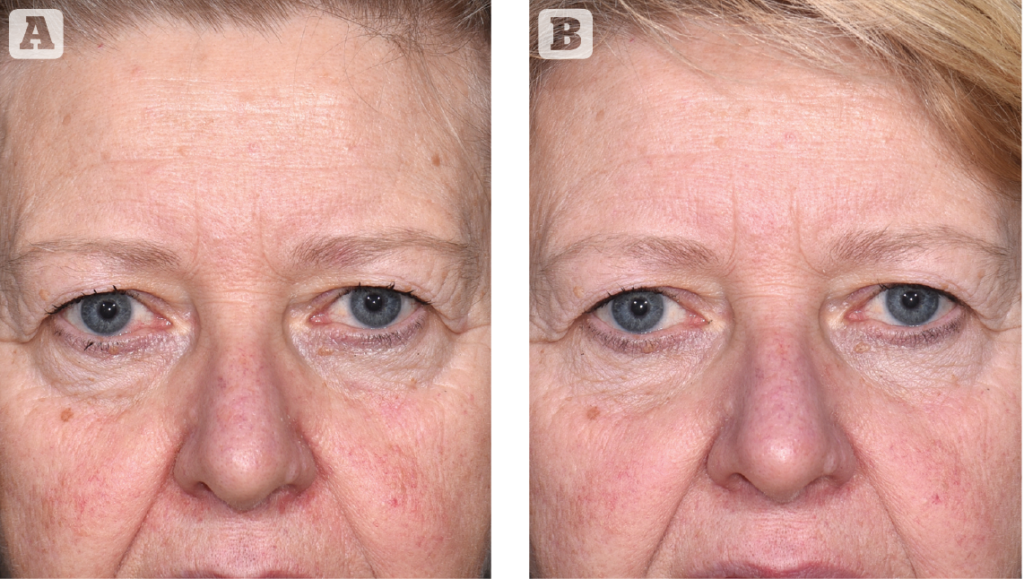
Agarose is suitable for increasing projection and support and for diffuse filling. It gets surrounded by histiocytes within hours after injection. This may help to maintain projection and support. The tissue response may become palpable and visible if concentrations higher than 1% get into contact with eyelid dermis or 1.5% with other dermis. The response to deeply injected 3.5% is sometimes palpable for several weeks. Reactive oedema immediately after injection can be pronounced, but mid- or long-term oedema is very rare. This results in a high degree of predictability and precision for volumising injections (Figure 7). Taking the absence of any chemical modification and the complete biodegradation into account, agarose is a good option for large volume treatments (Figure 8).
Patient selection and counselling
The effects of facial volume correction can be less obvious than wrinkle filling or muscle modulation. Not long after treatment, patients can genuinely believe that all the fat4 or all the filler20 is gone, while the clinical photography evidently shows the opposite.
In cases when pigmentation problems, wrinkles, muscle hypertonia or hypercontraction are significant, treating the obvious first may inspire more patient confidence than imposing a ‘from-the-basement-to-the-roof’ philosophy.
What patients see in the mirror can bother them to a larger degree than how others see them from all angles. We are getting disproportionate requests about tear troughs, the nose or the lips while lateral components of a balanced strategy may be refused. Patients not being bothered by their cheekbones is a poor reason for subcutaneous injection medial to a nasolabial fold. Excessive anterior fullness can point the patient out as having been injected, and the risk of intravascular injection is higher.
The high price of full-face volumising injections can take patient dissatisfaction to a different level than after wrinkle treatment.
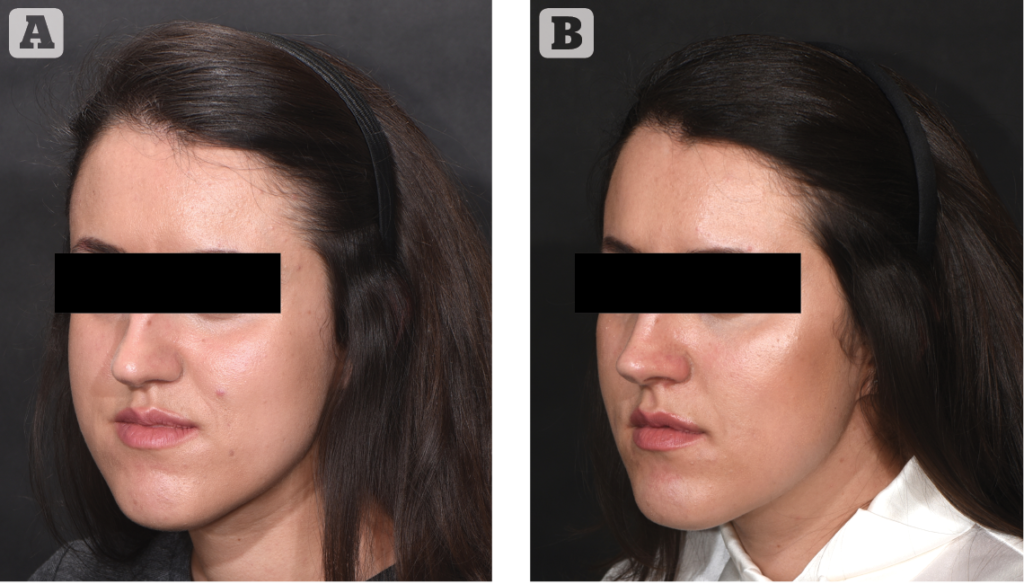
Therefore, proper informed consent about volumising injections requires considerable time and dedication. Injecting the temples, the eyebrows, the midface and the jawline with physiological saline solution (or a low concentration of local anaesthetic6) on one side is a great way to show a patient what can be achieved. This is not recommended for the forehead, the orbital margins, the nose or the lips.
Typical small versus large volume treatments and levels of injection
Treatments on the orbital margin, of the nose, the lips or the chin (Figure 9) rarely require more than 1 to 1.4 ml. If more volume is needed, it can be wise, before injecting more, to evaluate the result after three weeks, when all swelling caused by the trauma of injecting is gone and asymmetries and imperfections of the first treatment are more apparent.
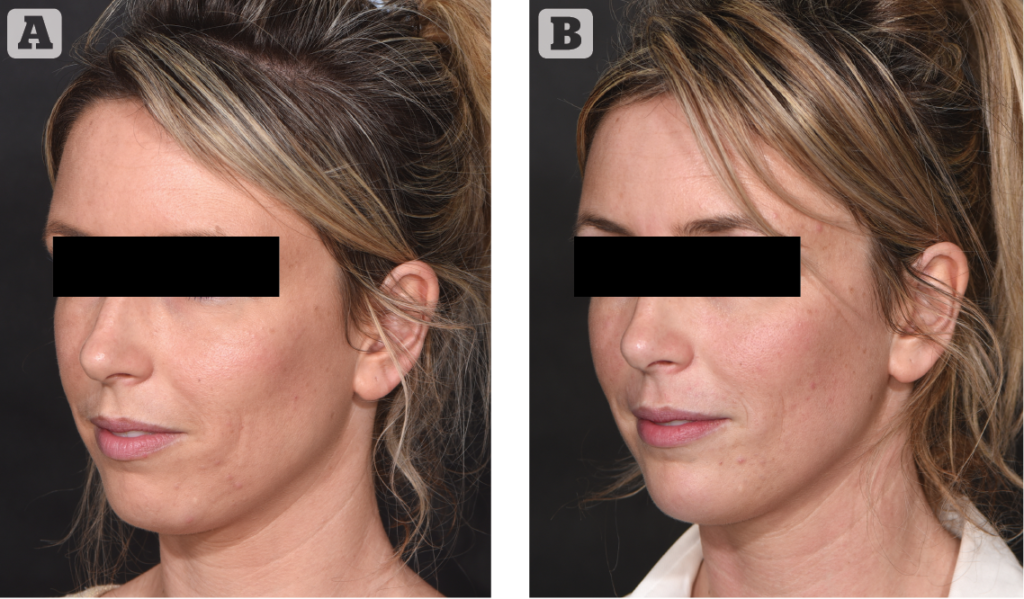
It is not uncommon for tens of millilitres of fat to be used in global facial rejuvenation by lipofilling. Although a part of the volume of injected fat disappears, one can still not expect to match the effect of global facial lipofilling by injecting only a few syringes of fillers. It is laudable that the maximal effects of minimal quantities are shown during clinical demonstrations. Nevertheless, immediate results are the sum of the injected volume and the swelling caused by the trauma of injection and remodelling. This ‘workshop effect’ may lead to overestimation of what can be achieved with a little quantity of filler.
Imperfections, such as hardening or irregular results, are better hidden after injection close to bone. Subcutaneous injections tend to give more visible results for less quantity. In most areas, injection close to bone holds less risk for intravascular injection than subcutaneous administration, while the latter, unjustly, may look less frightening to the inexperienced injector. Deep injections may be better at compensating lack of bony support. Extra superficial volume adds weight, which may pose a problem in descending soft tissue, e.g. close to the jowls. However, a subdermal injection may offer other benefits than volume augmentation. The gentle biostimulatory effect of calcium hydroxyapatite can improve skin firmness.
More therapeutic modalities for tightening and lifting have become available. Subdermal induction of heat, focused ultrasound, and suspension sutures require passage through the superficial compartments or target the superficial musculoaponeurotic system (SMAS). As we have very little data on interactions, it is wise to limit filler injections to deep and, for wrinkle filling, strictly intradermal if other subcutaneous treatments have been performed or are considered (Figure 10).
Variability of results and mid- to long-term oedema
As biostimulatory agents depend on tissue response, 3 to 4-month pictures are a fairer representation of the result than what is seen immediately or only a few days after injection. If the result of a small treatment looks pleasing in the mid-term, the use of larger quantities in that individual is more justified.
The low cellular response to HA fillers makes them more versatile for all levels of depth in facial rejuvenation. Soft lumps can be remodelled to some extent, while cellular aggregates or capsules cannot. The downside of HA is that the degree of reactive focal, local and locoregional oedema is hard to predict. Signs and symptoms of facial oedema, such as malar mounds or variable size of baggy eyelids throughout the day, and previous reactive swelling after HA injection, are relative contraindications, at least in and around the affected areas.
A little oedema may not be taken as a disadvantage by patients with relatively tight skin or those who revel in a slightly exaggerated lip profile. They may appreciate it as more result per volume. Nevertheless, injection of the lips, on the inferior orbital margin or superficially in the temples and the use of large quantities, in general, hold the risk of an oedematous, overinflated and unnatural look in some patients, especially those with lax soft tissues and thin skin. It is highly recommended to divide large volume HA treatments into multiple sessions, with intervals of at least three weeks. Clinical photography at 3 months and later is very helpful in detecting light puffiness and allowing the practitioner to act or abstain from action accordingly.
Agarose allows us to modify a face in a predictable manner with the injected volume and only that. Experienced patients need to be advised that no reactive oedema also means no more volume than the quantity injected. Great care has to be taken to avoid the formation of visible or palpable nodules when concentrations that are too high are deposited in contact with the dermis.
Due diligence in education
Sound knowledge of the anatomy, of aesthetic principles including age-, gender-, ethnicity- and culture-related changes, of injection techniques and safety are a prerequisite for volumising injections.
While intradermal injections in the vicinity of pore and adnexal microbiomes appear to be more forgiving, acute and low-grade subcutaneous or deep infections can have catastrophic consequences. After proper cleaning and preparing of the skin, maintaining the sterility of cannulas, needles and products cannot be forgone. Window-dressing with sterile drapes around a field that includes unprepared zones, or with sterile gloves for handling non-sterile equipment is not helpful.
It is easiest to reach anatomical points precisely with sharp needles. They cause less pain than cannulas when penetrating thick muscles (temporal, masseter, depressor labii inferioris). Cannulas make it easier to follow anatomical planes, such as between the superficial and muscle temporal fascia or under the orbicularis oculi muscles. If their calibre is 25 G or larger, their safety profile is better21. Because severe complications are rare, statistical evidence for preventive measures is hard to find, if at all. Nevertheless, knowledge of the different safety profiles of the fillers, the tools to inject them, and the preventive measures mentioned earlier11, help us to base injection techniques on reason rather than on intuitive preference.
Academic training programmes are evolving, but much of the knowledge about volumising injections is gathered at congress lectures and demonstrations. We would not stand where we are without training and workshops organised by the industry. More and more detailed descriptions of injection techniques18,20,22 are appearing in peer-reviewed literature. No single product can cater to all needs and practitioners are recommended to build knowledge across brands and companies and make their choices independently.
Cadaver courses offer great opportunities to improve knowledge about anatomy. Seeing the structures that we inject helps to understand the effects of our actions in clinical practice, although the vascular detail as exposed by a professional anatomist after the injection of intra-arterial resin will not be found. While we hope to work at precise levels and follow anatomical planes, it can be humbling to discover to what extent filler is deposited in adjacent structures (Figure 3).
Observational learning is different from personally injecting and dissecting. For proper dissection, tedious tasks have to be taken at heart. Raising the skin at the immediate, subdermal level or raising superficial fat flaps following anatomical planes can take a lot of time before any proper results can be seen. Deep, exploratory incisions without sufficient exposure lead to flipping tissue back and forth, thereby squeezing oil from the fat compartments, to messing up the dissection field, and frustration.
For supervised injection and dissection of all injectable areas of a face by two participants, alternating with lectures about the anatomy and clinical applications, at least two days are required, three are better. It may help to clearly differentiate in course programmes between observation and the opportunity to completely dissect the result of one’s own injections, including the effort it takes.
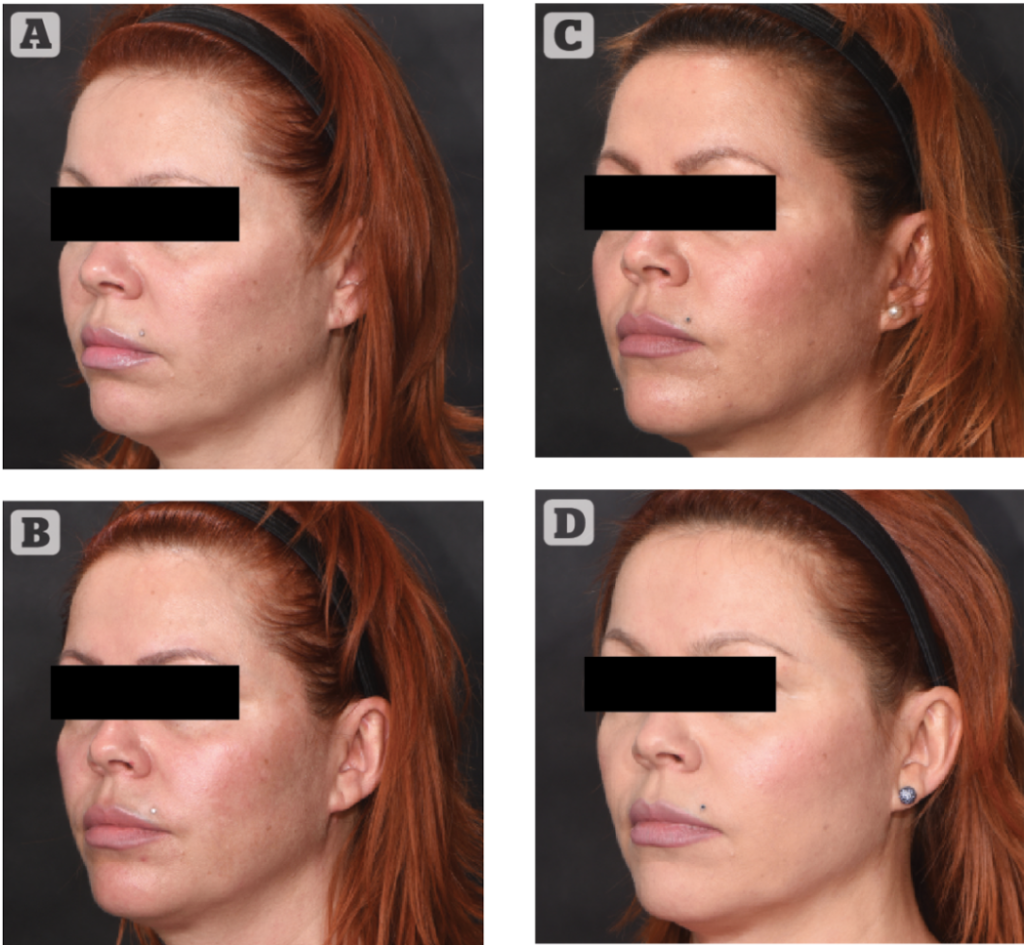
Clinical observation and hands-on training are particularly important to develop practical skills. It is helpful for trainers to grade anatomical targets by difficulty and risk. Deep needle injection of the temples or the chin are not difficult and carry less risk of intravascular injection. Cannula injection of the retro orbicularis oculi fat pad (ROOF) under the tail of the eyebrow, or of the nasolabial subcutis, is not difficult but carries a relatively high risk of catastrophe in case of intra-arterial injection. Orbital margin, nose and lip injections are difficult and carry a higher risk. Furthermore, protection of retinal circulation by external compression of collaterals can be arranged for nasal or ROOF treatment, not for A-frame or inferior orbital margin enhancements.
Research and development
Magnetic resonance imaging is becoming less expensive and calculating volume change from 3D photography more refined. Quantified evidence about mid- and long-term results would offer more than immediate results in demonstrations, selected clinical photography and patient satisfaction scores.
Remarkably few histologic reports have been published about filler behaviour and tissue response outside the scope of intradermal injections or filler complications. As muscles, fat compartments and connective tissue are being injected, such data would be welcome.
Automated injection devices, delivering precise micro-boluses or gentle, continuous flow, with a safety stop triggered by high-pressure gradients, are already at hand23,24. They can prevent us from exerting too much pressure to unblock a clogged-up needle, or from injecting too fast after a pressure drop on entering a blood vessel. It would greatly contribute to safety if every injection could be preceded by automated aspiration.
The smaller the calibre of a needle, the smaller the bolus that can be injected over a given amount of time, even under relatively high pressure. This may help to reduce the consequences of inadvertent intravascular injection. The evolution towards volumising fillers that can pass a 30 G needle has been a step forward for safety and remains a good standard for new developments.
The only terminal arteries in the face are the central retinal arteries. The only clinically documented compartment syndrome in the face is a retrobulbar hematoma. For injections to cause necrosis in any other area of the face, external pressure or segmental embolisation of an artery is (fortunately) insufficient. The block has to extend into the capillaries because collateral arteries otherwise maintain perfusion. How progressive, intravascular coagulation and inflammation contribute to the ischemia deserves further attention. Skin appearance after embolisation does not necessarily go from pallor to livedo reticularis9 as described by DeLorenzi. Clinical pictures on HA embolisation cases show bright red zones containing small foci of necrosis25,26. Both the redness and gradual progression into adjacent zones26 are indications of an expanding inflammatory process, leading to focal, intravascular coagulation. Laboratory studies comparing what happens after intravascular injection of different fillers would be most welcome.

Conclusions
Over the last decades, we have witnessed a multiplication of anatomical knowledge relevant to volumising treatments. A large array of injectables are now available, including volumising fillers, biostimulants, and combinations. Indications are growing in numbers.
Reasonably extensive information has been published about tolerance, safety, patient satisfaction and rare but severe complications. Histology of filler behaviour and tissue response is scarce, as are quantified data about volume increase per unit of injectable and resorption over time.
Volumising treatments are of three-dimensional complexity without the control of surgical exposure. Obtaining informed consent requires more time and effort than for muscle modulation and wrinkle filling. Peer-reviewed literature on injection techniques, hands-on training and cadaver courses help to progress from basic treatments to complex injections and those with more inherent risk of serious adverse events.
Declaration of interest Dr. Vandeputte received no payment for writing this article.
Dr. Vandeputte is a consultant and trainer for Merz Pharma Merz Pharma GmbH & Co. KGaA, Frankfurt am Main, Germany and for Advanced Aesthetic Technologies Inc, Brookline MA, USA (AAT).
The dissections show in this article were performed at the Cadaveric Microanatomy Centre, Applied Science University, Amman, Jordan and were funded by AAT, as part of the Algeness Inject and Dissect Course, 27-29 August, 2019, chaired by the author.
Figures 1–10 © Dr Vandeputte
* Belotero Volume and ****Radiesse are registered trade names of Merz Pharma Merz Pharma GmbH & Co. KGaA, Frankfurt am Main, Germany
** Restylane and **** Sculptra are registered trade names of Galderma, Lausanne, Switzerland
*** Algeness is a registered trade name of Advanced Aesthetic Technologies Inc, Brookline MA, USA
****** Attiva is a registered trade name of Tema Medicina, Bologna, Italy
REFERENCES
- Coleman SR, Grover R. The anatomy of the aging face: Volume loss and changes in 3-dimensional topography. Aesthetic Surg J. 2006;26(1 SUPPL.).
- Fitzgerald R, Graivier MH, Kane M, Lorenc ZP, Vleggaar D, Werschler WP, et al. Update on Facial Aging. Aesthetic Surg J. 2010;30(1_Supplement):11S-24S.
- Fitzgerald R, Graivier MH, Kane M, Lorenc ZP, Vleggaar D, Werschler WP, et al. Nonsurgical Modalities to Treat the Aging Face. Aesthetic Surg J. 2010;30(1_Supplement):31S-35S.
- COLEMAN SR. Structural Fat Grafting. St. Louis, MO: Quality Medical Publishing; 2004.
- Fraser JR, Laurent TC, Laurent UB. Hyaluronan: its nature, distribution, functions and turnover. J Intern Med. 1997;242(1):27–33.
- Lambros V. Volumizing the Brow With Hyaluronic Acid Fillers. Aesthetic Surg J. 2009;29(3):174–9.
- Micheels P, Besse S, Vandeputte J. Cohesive Polydensified Matrix® cross-linked hyaluronic acid volumizing gel: a magnetic resonance imaging and computed tomography study. Clin Cosmet Investig Dermatol [Internet]. 2018 Dec;Volume 12(12):1–10. Available from: https://www.dovepress.com/cohesive-polydensified-matrixreg-cross-linked-hyaluronic-acid-volumizi-peer-reviewed-article-CCID
- Ozturk CN, Li Y, Tung R, Parker L, Piliang MP, Zins JE. Complications following injection of soft-tissue fillers. Aesthetic Surg J. 2013;33(6):862–77.
- Delorenzi C. Complications of injectable fillers, Part 2: Vascular complications. Aesthetic Surg J. 2014;34(4):584–600.
- Beleznay K, Carruthers JDA, Humphrey S, Jones D. Avoiding and treating blindness from fillers: A review of the world literature. Dermatologic Surgery. 2015.
- Beleznay K, Carruthers JDA, Humphrey S, Carruthers A, Jones D. Update on avoiding and treating blindness from fillers: A recent review of the world literature. Aesthetic Surg J. 2019;39(6):662–74.
- American Society for Aesthetic Plastic Surgery A. The American Society for Aesthetic Plastic Surgery’s Cosmetic Surgery National Data Bank: Statistics 2018. Aesthetic Surg J [Internet]. 2019 Jun 21;39(Supplement_4):1–27. Available from: https://academic.oup.com/asj/article/39/Supplement_4/1/5521858
- Beleznay K, Carruthers JDA, Carruthers A, Mummert ME, Humphrey S. Delayed-Onset Nodules Secondary to a Smooth Cohesive 20 mg/mL Hyaluronic Acid Filler: Cause and Management. Dermatologic Surg. 2015;41(8):929–39.
- Cotofana S, Gotkin RH, Frank K, Koban KC, Targosinski S, Sykes JM, et al. The functional anatomy of the deep facial fat compartments: A detailed imaging-based investigation. Plast Reconstr Surg. 2019;143(1):53–63.
- Pilsl U, Rosmarin W, Anderhuber F. The premaxillary space: A location for filler injection? Dermatologic Surg. 2014;40(3):301–4.
- Pavicic T, Mohmand HM, Yankova M, Schenck TL, Frank K, Freytag DL, et al. Influence of needle size and injection angle on the distribution pattern of facial soft tissue fillers. J Cosmet Dermatol. 2019;18(5):1230–6.
- Vandeputte J, Leemans G, Dhaene K, Forsyth R, Vanslembrouck J, Hatem F, et al. Spreading Pattern and Tissue Response to Hyaluronic Acid Gel Injections in the Subcutis: Ultrasound Videos, Ultrasound Measurements, and Histology. Aesthetic Surg J [Internet]. 2020 Jan 17;0954162(478):1–4. Available from: https://academic.oup.com/asj/advance-article/doi/10.1093/asj/sjaa007/5707605
- Mckee D, Remington K, Swift A, Lambros V, Comstock J, Lalonde D. Effective Rejuvenation with Hyaluronic Acid Fillers : Current Advanced Concepts. Plast Reconstr Surg. 2019;1277e-1289e.
- Lambros V. A technique for filling the temples with highly diluted hyaluronic acid: The “dilution solution.” Aesthetic Surg J. 2011;31(1):89–94.
- Vandeputte J. Real-world Experience with Volume Augmentation using Cohesive Polydensified Matrix Hyaluronic Acid Gel: A Retrospective Single-center Analysis of 110 Consecutive Patients with Medium- to Long-term Follow-up. J Clin Aesthet Dermatol [Internet]. 2018;11(12):30–9. Available from: http://jcadonline.com/hyaluronic-acid-dec-2018/
- Humzah MD, Ataullah S, Chiang CA, Malhotra R, Goldberg R. The treatment of hyaluronic acid aesthetic interventional induced visual loss (AIIVL): A consensus on practical guidance. J Cosmet Dermatol. 2019;18(1):71–6.
- Romeo F, Romeo F. Upper Eyelid Filling Approach [ U . E . F . A .] Technique : State of the Art After 500 Consecutive Patients. Aesthetic Plast Surg [Internet]. 2019;43(3):663–72. Available from: https://doi.org/10.1007/s00266-018-1296-6
- Sundaram H, Rohrich RJ, Liew S, Sattler G, Talarico S, Trévidic P, et al. Cohesivity of hyaluronic acid fillers: Development and clinical implications of a novel assay, pilot validation with a five-point grading scale, and evaluation of six US food and drug administration-approved fillers. Plast Reconstr Surg. 2015;136(4):678–86.
- Dugaret AS, Bertino B, Gauthier B, Gamboa B, Motte M, Rival Y, et al. An innovative method to quantitate tissue integration of hyaluronic acid-based dermal fillers. Ski Res Technol. 2018;(January):423–31.
- Ciancio F, Tarico MS, Giudice G, Perrotta RE, Lemperle G. hyaluronidase use in preventing skin necrosis after treatment with dermal fillers : Report of two cases [ version 2 ; peer review : 2 approved , 1 not approved ] Referee Status : 2019;1–11.
- Wibowo A, Mohan K. Reversal of Post-filler Vision Loss and Skin Ischaemia with High- Dose Pulsed Hyaluronidase Injections. Aesthetic Plast Surg [Internet]. 2019;43(5):1337–44. Available from: https://doi.org/10.1007/s00266-019-01421-6