Abstract
The 1927 nm wavelength handpiece (Clear + Brilliant Perméa) is a new addition to the Clear + Brilliant laser system, which also includes the original 1440 nm handpiece, and is a low-level non-ablative fractional laser that is FDA-cleared for general skin resurfacing1. The 1927 nm wavelength confers an increased ability to target dyschromia and superficial processes associated with photodamage. A post-market surveillance study was conducted to determine clinical efficacy, overall patient satisfaction, and skin responses to laser treatment in conjunction with a topical skincare regimen at nine centres across the United States. Subjects received six laser treatments at 2-week intervals. Seventy-eight percent of patients reported noticeable improvement in skin tone or texture 2 weeks after the initial treatment. Eighty-six percent of subjects reported being satisfied to very satisfied with the overall treatment series 2 weeks after the sixth treatment visit. Treatments were well-tolerated, with minimal erythema and oedema lasting no longer than 24 hours. One patient experienced post-inflammatory hyperpigmentation after the third treatment, with no other adverse events or side-effects reported. In summary, the Clear + Brilliant Perméa laser treatment, combined with SkinCeuticals skincare, resulted in improvements in skin tone and texture, while maintaining a high level of patient satisfaction.
The Clear + Brilliant Perméa System (Solta Medical, Inc., Hayward, CA) is a low-level non-ablative fractional laser that resurfaces skin to a maximum depth of 170 µm with maximum treatment coverage at 5% using a 1927 nm handpiece. This novel 1927 nm wavelength is an addition to the previously FDA-cleared Clear + Brilliant laser system, which consisted of the original 1440 nm wavelength handpiece only1. While the Clear + Brilliant system is FDA-cleared for general skin resurfacing1, the 1927 nm wavelength handpiece has an increased ability to target dyschromia and superficial processes associated with photodamage. The 1927 nm wavelength confers a higher absorption coefficient for water, thus targeting superficial processes associated with photodamage2–4, with greater capacity than the 1440 nm wavelength.
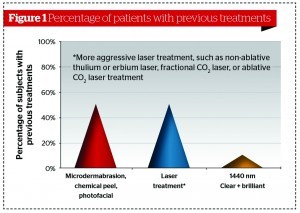
A post-market surveillance evaluation, conducted from May 2012 to December 2012, assessed clinical efficacy, overall patient satisfaction, and skin responses to laser treatment in conjunction with a topical skincare regimen at nine independent centres across the United States. This article summarises the clinical findings and results of 78 patients, Fitzpatrick skin types I–V, who enrolled to undergo a series of six laser treatments spaced 2 weeks apart. In conjunction with Perméa laser treatment, patients were given skincare products (SkinCeuticals, Inc., Garland, TX) to be used during and after the laser treatments. Specifically, SkinCeuticals CE Ferulic® serum (topical 15% L-ascorbic acid, 1% alpha tocopherol, and 0.5% ferulic acid), a topical antioxidant at pH 3.2 was used during and immediately post-laser treatments, and in the intervals between laser treatments during the course of the entire series. Prior ex vivo studies using forearm skin biopsies demonstrated increased pigment removal with the 1927 nm laser treatment in combination with CE Ferulic serum in comparison with untreated controls, laser only, and CE Ferulic serum only5.
The Clear + Brilliant Perméa (1927 nm) handpiece differs from its more powerful sister device, the 1927 nm wavelength associated with the Fraxel® Dual 1550/1927 Laser System (Solta Medical, Inc., Hayward, CA), in that it is a low-power 1 Watt laser versus the high-power 10 Watt Fraxel Dual Laser System. Another key difference is in beam profile, where the Fraxel Dual’s 1927 fibre laser has a pure TEM00 Gaussian, while the diode laser in Perméa has a more flat-top profile. The pulse energy range is fixed at 5 mJ for Perméa in contrast to a range of 5–20 mJ for the Fraxel Dual Laser System2–4. Thus, for Perméa, there is no topical numbing required and no downtime. Many of the nine treatment sites represented in this article have safely delegated the Clear + Brilliant Perméa treatment to a clinical aesthetician or nurse to perform the treatment, or as appropriately designated in state/country regulations.