Bioengineering injectable biomaterials for soft tissue fillers
Most of the currently available HA injectable biomaterials used in soft tissue fillers consist of particles (a solid phase) suspended in a fluid phase18. The physicochemical structure of this soft tissue filler is established during the manufacturing process by the adjustment of a number of variables including, but not limited to:
- Concentration of the solid-phase particles
- Method and percentage of cross-linking of the solid‑phase particles
- Type and technology of cross-linker used
- Proportion of gel in the fluid phase (gel-to-fluid ratio).
HA soft tissue fillers with different physicochemical properties produce different clinical outcomes with regard to their rheology — elasticity and viscosity18. One important rheological property of a soft tissue filler gel that can be quantified is its elastic modulus (G’). A high G’ in a HA soft tissue filler appears to be a predictor of a better resistance to skin tension forces; therefore, it deforms less under pressure18.
The rheological properties can be used as a scientific rationale in choosing a soft tissue filler — a strategy known as rheological tailoring18. This can be individualised for each patient and facial area to achieve the desired goals and outcomes. To that end, viscosity and elasticity should be part of the selection process when choosing an appropriate soft tissue filler. Other clinical considerations, such as injection technique and implantation depth of soft tissue filler, are also important18. The addition of B. subtilis-derived HA to aesthetic medicine practitioners’ armamentarium can increase their ability to achieve customised, evidence-based outcomes from a rheological perspective.
Potential complications associated with injectable HA biomaterials
As with every medical procedure, there is a degree of risk associated with the use of injectable HA soft tissue fillers, although serious complications arising from their use are infrequent19. Unwanted adverse events do occur with all soft tissue filling compounds (including HA biomaterials); however, their true prevalence is unknown4, 20–22. These adverse events may be injection‑related, technique‑related, or (rarely) may be owing to localised exposure to HA itself, potential residual purification solvents, or trace presence of endotoxin. Injection-related events are those that are caused by the injection of the soft tissue filler rather than the soft tissue filler itself, while technique-related events are the result of the specific manner in which a physician injects the substance into the patient.
Injection-related adverse events
By far the most common adverse events associated with HA soft tissue fillers are injection-related19. These events are usually transient, resolving within 4–7 days, and are localised to the site of injection (Table 2). Rarely, an inadvertent intravascular injection or adjacent vascular compression may result in a non-localised adverse event (i.e. necrosis)19, 23.
Technique-related adverse events
One of the most common technique-related adverse events is inappropriate placement of the soft tissue filler. Lumps of visual product or bluish bumps under the skin (the Tyndall effect) can result from a too superficial placement of product24, 25. For the most part, such reactions can be prevented by the use of correct injection technique and proper training on the part of the injector19. The occurrence of these events can lead to anxiety, dissatisfaction, and less than optimal results for the patient24,25. The advantage of HA-based soft tissue fillers over other classes (e.g. Poly-L-lactic acid, Calcium hydroxylapatite) is their reversibility when treated with hyaluronidase, which can successfully resolve many unwanted adverse events20, 25.
Serious adverse events related to HA
Injectable HA soft tissue fillers are typically well tolerated with few adverse events. Serious events are possible, although rare. Typically these events have not been associated with aesthetic use of HA products.
HA-derived injectable soft tissue fillers may rarely be associated with localised reactions, such as persistent swelling, pain, and nodule formation. These effects may require physical removal and/or enzymatic degradation with hyaluronidase4, 19. Other rare effects include angioedema, arterial occlusion, loss of vision, infection, necrosis, vasculitis and vascular occlusion23, 26–31. Practitioners should be able to recognise these adverse events and apply the appropriate clinical algorithm for treatment should they occur9, 31–33.
Conclusions
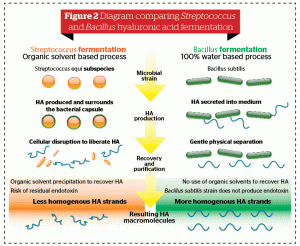
A primary challenge for manufacturers of soft tissue fillers has been obtaining HA of high quality and purity. Contrary to Streptococcus-derived HA, B. subtilis-derived HA is produced by a host, free of endotoxin and without organic solvents.
The B. subtilis-derived HA process overcomes the manufacturing and safety challenges associated with HA of other origins, allowing for a high level of purity and a homogeneous end-product. The bioengineering process associated with production of B. subtilis-derived HA does not require the use of powerful organic solvents to extract it from the bacterial capsule, in contrast to the process required for Streptococcus-derived HA. In addition, B. subtilis-derived HA results in uniform strands and has a homogenous molecular weight with a narrow and well-defined weight distribution. In summary, B. subtilis-derived HA is well placed in the market to complement existing sources of HA used in soft tissue fillers.