Ding Wang, Carolie Horvath and Susan D. Lewis discuss the results of their open-label, dose-ranging study on the use of Essential Eyebrow Solution for protection against eyebrow loss.
One of the hallmark characteristics of cancer cells is that they exhibit rapid cell division and proliferation. Chemotherapy treatment targets all rapidly dividing cells by damaging cell structures or metabolism. Chemotherapeutic agents impair mitotic and metabolic processes of actively growing hair follicles, causing a weakening of the partially keratinized, proximal portion of the hair shaft, which results in thinning of the hair shaft, making it fragile and susceptible to breakage1,2. This disruption of the hair growth cycle causes alopecia, a term which encompasses any loss of hair, not only from the scalp, but from other areas of the body, including the eyebrows.
There are four major classes of chemotherapeutic agents most commonly responsible for chemotherapy-induced alopecia, with the extent of induced hair loss differing across the classes. The classes are: antimicrotubule agents (e.g., paclitaxel), topoisomerase inhibitors (e.g., doxorubicin), alkylators (e.g., cyclophosphamide), and antimetabolites (e.g., 5-fluorouracil plus leucovorin)3. Drugs with high potential for inducing alopecia include adriamycin, cyclophosphamide, daunorubicin, docetaxel, epirubicin, etoposide, ifosphamide, irinotecan, paclitaxel, topotecan, vindesine, and vinorelbine. Paclitaxel typically induces complete hair loss, including both the scalp and eyebrows. Adriamycin induces complete hair loss on the scalp, and very often causes eyebrow hair loss. Chemotherapeutic agents such a methotrexate, cytoxan, carboplatin, and 5-fluorouracil can cause hair loss in some patients but not others. Eyebrow hair loss does not occur with all chemotherapeutic agents, but depends on the specific chemotherapy regimen used, length of treatment, and patient sensitivity to the side-effects of a specific agent.
It is estimated that 65% of the patients who undergo chemotherapy experience overall hair loss3, yet more than 80% of cancer patients who receive chemotherapy consider hair loss to be the worst aspect of their chemotherapy experience, and 8% of female cancer patients say they would even decline treatment for fear of this side-effect3,4. For many men, hair loss during chemotherapy negatively affects their self-perception of virility and masculinity. The authors find that it is not uncommon for a chemotherapy patient to base their decision on which chemotherapy regimen to choose, when an option exists, on the likelihood of alopecia as a side-effect of treatment.
Eyebrow hair loss results in cosmetic changes that can cause emotional distress in many cancer patients. The eyebrows are one of the most important features of the face. Along with the eyelashes, they protect our eyes from sweat, dust, and debris. Additionally, they create a frame for the face, enhance attractiveness, and aid non‑verbal communication. Eyebrows contribute significantly to the expression of emotions. Massachusetts Institute of Technology behavioral neuroscientist Javid Sadr has theorized that, despite the many evolutionary changes humans have undergone, eyebrows have remained on our faces because they are crucial to facial identification. In 2002, he and his colleagues conducted a study in which volunteers were asked to identify fifty famous faces. The photos were digitally altered to show faces without eyebrows or without eyes. When celebrities lacked eyes, subjects could recognize them nearly 60% of the time. However, when celebrities lacked eyebrows, subjects recognized them only 46% of the time. As Sadr points out, eyebrows stand out against the backdrop of your face, and therefore, not only identify who you are but how you’re feeling5.
Although cranial prostheses (wigs) and other forms of 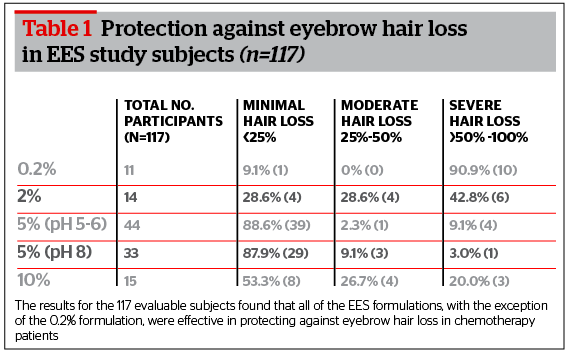
Essential Eyebrow Solution (EES) is a specific formulation of black cohosh compounded to address eyebrow hair loss. Used for many medicinal purposes, black cohosh (Cimicifuga racemosa) is a perennial herb native to North America with a large, creeping rhizome. Its name comes from the Latin words cimex and fugo meaning ‘to drive away bugs’, referring to its use as a vermifuge. Native Americans used black cohosh to treat uterine disorders associated with menstrual and menopausal symptoms, as well as pain during childbirth, and rattlesnake bites. The herb has been commonly prescribed in European countries, especially during the 1950s, as hormone replacement therapy during menopause7.
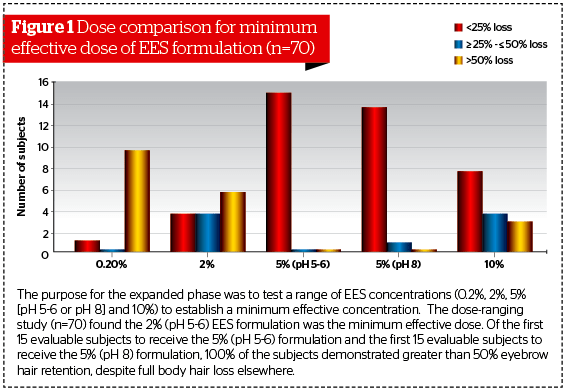
The EES formulation was developed when it was found that extracts from black cohosh were a common ingredient in certain compositions of hair loss remedies. Because there was no standardized utilization or application for this use of black cohosh, the safety and efficacy of EES was established in a preliminary, open-label, dose-ranging study of chemotherapy treatment subjects.
Patients and methods
Products studied
The patented topical EES formulations used during the study were compounded by a research laboratory under good manufacturing practices. EES is composed of black cohosh and a mild surfactant, which is diluted in water for rapid absorption and drying. It also contains standard preservatives, as well as citric acid (pH adjuster). The formulations were developed in 5% (pH 5–6) and 5% (pH 8) concentrations for the initial phase of this study and 0.2% (pH 5–6), 2% (pH 5–6), and 10% (pH 5–6) concentrations for the expanded phase. A pH of 5–6 was chosen for the expanded phase based on the preliminary results from the initial study phase, and the fact that this pH is more compatible with the skin. The solution was packaged in light-resistant two-ounce (59.147 ml) resin containers; at room temperature storage EES is safe and effective for up to 2 years.
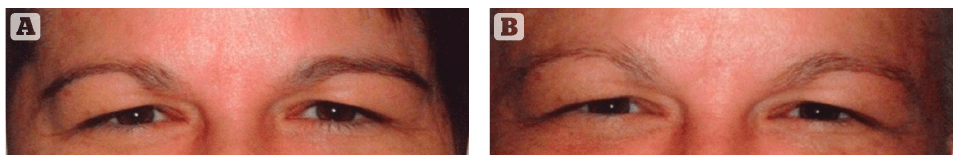
Figure 2 (A) Subject #58 before applying the 5% (pH 5-6) EES formulation, prior to undergoing 12 weeks of chemotherapy consisting of Taxotere and Cytoxan for breast cancer. (B) Subject #58 experienced full body hair loss with the exception of the eyebrows, which were retained after chemotherapy.
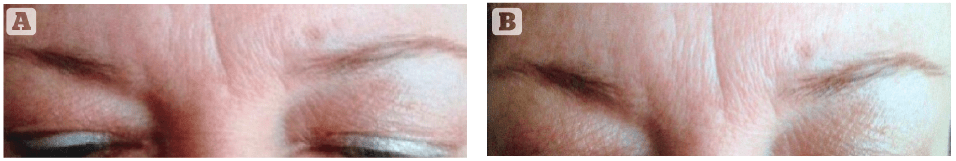
Figure 3 (A) Subject #73 before applying the 5% (pH 5-6) EES formulation, prior to undergoing 18 weeks of chemotherapy, consisting of Taxotere and Cytoxan for breast cancer. (B) Subject #73 applied the 5% (pH 5-6) EES formulation, while undergoing 18 weeks of chemotherapy, consisting of Taxotere and Cytoxan for breast cancer, retaining 100% of her eyebrow hair, despite full body hair loss elsewhere.
Pre-study product safety testing
Because use of EES requires multiple applications to the skin, the 5% (pH 5–6) formulation was tested for skin sensitivity to determine safety for topical use. A repeat insult patch test (RIPT) was conducted on 50 healthy male and female human subjects, with an age range of 18–68 years. The RIPT was conducted under good clinical practices with an attending dermatologist and institutional review board (IRB) approval. The results of the RIPT demonstrated that EES was a non-primary irritant and non-primary sensitizer to the skin. In addition, a choriollantoic membrane vascular assay (CAMVA–14 Day), a bovine corneal opacity and permeability test (BCOP), and a sterility test were conducted by an accredited laboratory, with passing results.
Study objectives
The objectives of the study protocol were to assess the safety and efficacy of EES when administered to cancer subjects receiving intensive chemotherapy regimens. The primary endpoints aimed to determine the following:
- The percentage of subjects experiencing minimal (<25%), moderate (25–50%), or severe eyebrow hair loss (>50%) as a result of chemotherapy treatment while using EES
- The percentage of subjects experiencing dermatologic adverse events related to use of EES
- The number of dermatologic usage-related adverse events experienced by study subjects.
A secondary objective was to determine the most effective concentration of EES for the protection against or reduction of eyebrow hair loss in subjects receiving various chemotherapy agents. In order to help achieve these study objectives, as well as to ensure that subject safety, welfare, and rights were protected, various legal and regulatory affairs experts were consulted to certify that all appropriate measures were in place to satisfy requisite rules and regulations related to the study of EES. EES was developed as a botanical cosmetic for the purposes of this study and was therefore not subject to United States Food and Drug Administration (FDA) premarket approval. EES will be further developed as a botanical drug in the United States. This study was voluntarily undertaken to gather important proof of efficacy information.
Study subjects
One hundred and forty-nine subjects consented to the EES study participation. Of these 149 subjects, 143 were cancer patients with a diverse range of cancer diagnoses (including breast, ovarian, anal, bladder, endometrial, and lung cancers, as well as leukemia and lymphoma). The remaining 6 subjects were diagnosed with alopecia areata (non-cancer related) and, therefore, were not included in the final data analysis. The study subjects ranged in age from 18–81 years, with an average age of 55 years. There were 148 female subjects and 1 male subject.
Study enrollment was conducted over a 2½-year period. All 149 of the consenting subjects were enrolled in the study. Enrollment was defined as a consenting subject who was assigned to an application regimen, whether or not they went on to use EES and assess their progress. Of the 149 enrolled subjects, 143 (142 female, 1 male) were undergoing chemotherapy with various chemotherapeutic regimens that are known to cause chemotherapy-induced alopecia. Twenty-six enrolled subjects did not complete the study: five subjects chose to discontinue study participation prior to using the EES solution and the other twenty-one subjects failed to comply with study regimen due to changes in their physical and/or emotional health status.
An experienced hair replacement specialist obtained the informed consent of each study subject prior to their use of the EES formulation. Each subject was advised of the voluntary nature of study participation, study purpose, study procedures, potential benefits and risks to study participation, right to withdraw at any time, and anticipated time commitment. Potential risks listed in the informed consent included mild skin irritation and dryness of the eyebrows. Subjects were educated on the importance of contacting the study staff and their physician immediately if they believed they were experiencing any side-effects related to the use of EES. Additionally, subjects were cautioned to avoid contact to the eyelashes or eyes, and were given instructions on how to flush eyes if contact were to occur.
As part of the informed consent process, subjects were asked to allow before-and-after photos (2–4 weeks post study) to objectively measure the effect of EES. Eyebrow hair preservation assessments supported by the before-and-after photos of the study subjects were conducted by a hair replacement specialist representing the study sponsor, and subjects served as their own control. During the study, subjects were routinely contacted by telephone to determine the following:
- Subject’s overall physical condition
- Subject’s compliance to EES application regimen
- Any noticeable adverse side-effects related to the EES product
- Any noticeable eyebrow loss and/or other body hair loss.
Enrolled study subjects were assigned to receive the EES formulation in one of the following concentrations: 0.2%, 2%, 5% (pH 5-6), 5% (pH 8), or 10%. Study subjects were instructed to apply the EES formulation topically to their eyebrows twice daily (morning and evening) beginning one week prior to their first dose of chemotherapy. Subjects were directed to continue this application for 2 months after the completion of their chemotherapy.
Statistical methods
Of the 143 enrolled chemotherapy treatment subjects, 117 subjects completed the study and were analyzed using basic percentages to calculate eyebrow retention.
To evaluate effectiveness of EES in protecting against eyebrow hair loss, the use of ‘minimal hair loss’ was used to correlate with eyebrow hair loss of less than 25% (<25%). ‘Moderate hair loss’ assessments correlated with eyebrow hair loss of greater than or equal to 25% but less than or equal to 50% (>25% to <50%). ‘Severe hair loss’ correlated with eyebrow hair loss of greater than 50% (>50% to 100%).
The study had two phases — a preliminary phase that focused on safety, tolerability, and efficacy. This was followed by an expanded phase which was a dose-ranging study to determine the minimum, and optimum, effective dose. For the expanded phase, the dosage comparison was made using data from 70 subjects. In order to keep the different dose cohort sizes comparable, dose comparison focused on the first 15 participants enrolled in each 5% EES formulation group.
Results
A total of 117 enrolled chemotherapy treatment subjects (82%) completed the study and were evaluable for data analysis. Evaluable subjects were those who received 12–18 weeks of chemotherapy while concurrently applying the EES formulation as directed. Greater than half of the 117 subjects analyzed were breast cancer subjects; however, the EES formulation was highly effective when used by the non-breast cancer subjects with similar intensive chemotherapy regimens. Success was defined as eyebrow hair loss of 50% or less. The results for the 117 evaluable subjects found that all of the EES formulations, with the exception of the 0.2% formulation, were effective in protecting against eyebrow hair loss in chemotherapy patients (Table 1). EES was found to be successful in 91% or 40 out of 44 subjects in the 5% (pH 5–6) EES formulation group.
The purpose for the expanded phase was to test a range of EES concentrations (0.2%, 2%, 5% [pH 5–6 or pH 8] and 10%) to establish a minimum effective concentration. The dose-ranging study (n=70) found the 2% (pH 5–6) EES formulation was the minimum effective dose. Dose-response evaluations were based on data from all subjects assigned to the 0.2%, 2%, and 10% EES concentration groups. Only the first 15 subjects from both 5% EES formulations were included for the dose-ranging evaluation to ensure a more equal sample size comparison amongst all concentration groups. The variability in the number of subjects enrolled to receive each EES formulation was a result of the timing of enrollment (initial versus expanded phase). There was no pre-determined enrollment number established prior to the conduct of each study phase.
The 0.2% EES formulation which was used by 11 evaluable subjects was shown to be ineffective in protecting against eyebrow hair loss. Ninety-one percent of the subjects experienced full body hair loss, including eyebrows. Of the 14 evaluable subjects who had completed the study using the 2% EES formulation, 57% had greater than 50% eyebrow retention and 43% had 50-100% eyebrow hair loss, despite all 14 subjects experiencing full body hair loss elsewhere. Of the first 15 evaluable subjects to receive the 5% (pH 5–6) formulation and the first 15 evaluable subjects to receive the 5% (pH 8) formulation, 100% of the subjects demonstrated greater than 50% eyebrow hair retention, despite full body hair loss elsewhere. Of the 15 evaluable subjects utilizing the 10% EES formulation, 80% retained greater than 50% of their eyebrows and the remaining 20% experienced greater than 50% eyebrow hair loss, despite all 15 subjects experiencing full body hair loss elsewhere (Figure 1).
There were no subjects in the 0.2%, 2%, 5% (pH 5–6) and 10% EES study groups who reported adverse effects related to the use of the EES formulation. One subject in the 5% (pH 8) group reported minor itching in the eyebrow area, that report was not substantiated.
There are two sets of before-and-after photos provided below (Figures 2–3), which both show the retention of eyebrow hair in patients using the 5% (pH 5–6) EES formulation.
Subject #58 applied the 0.5% EES formulation, while undergoing 12 weeks of chemotherapy consisting of Taxotere and Cytoxan (Baxter Healthcare Corp., Deerfield, IL, USA) for breast cancer and experienced full body hair loss with the exception of the eyebrows, which were retained (Figure 2).
Subject #73 applied the 5% (pH 5–6) EES formulation, while undergoing 18 weeks of chemotherapy, consisting of Taxotere and Cytoxan for breast cancer, retaining 100% of her eyebrow hair, despite full body hair loss elsewhere (Figure 3).
These subject-specific results are representative of the majority of the subjects assigned to the same dosage group. Additionally, many EES study subjects who experienced positive results commented that their eyebrows seemed to be stronger, and that the EES formulation promoted suppleness and shine. The positive psychological impact of eyebrow hair retention (even 50% retention) was also noteworthy, as many of the EES subjects who were provided with a cranial prosthesis (wig) found that they were able to recreate their appearance prior to receiving chemotherapy.
Discussion
The preliminary EES study data met the stated objectives and demonstrated that the 5% (pH 5–6) EES formulation was safe and effective when administered for the purpose of eyebrow hair preservation in chemotherapy treatment subjects. The 5% (pH 5–6) EES formulation was proven to be the most effective formulation in protecting against eyebrow hair loss in 91% of subjects. The minimal effective concentration was found to be the 2% EES formulation.
Analysis of the cancer chemotherapy regimens administered to these subjects demonstrated that EES was effective in retaining the subject’s eyebrows, while they experienced full body hair loss elsewhere as a result of their chemotherapy regimens. Based upon the responses from study subjects, the EES formulation may also contribute to the strength, suppleness, and shine of the eyebrows.
Inherent limitations in this study include a predominantly female, Caucasian population from a relatively small geographic area. Therefore, further research could be designed to include a more diverse subject population. Additionally, tighter controls on permitted chemotherapy regimens, monitoring of subject compliance with treatment administration, and monitoring of proper product application could be beneficial to the study of the efficacy and safety of EES.
Conclusion
Chemotherapy-induced alopecia reduces the psychological well-being of cancer patients because hair loss is a visible sign of cancer and treatment. Eyebrows are important to the countenance, appearance of vitality, and expression of emotions, and their loss often has a notable negative impact on a cancer patient’s psychological state. Stress-related psychosocial factors may have an adverse effect on cancer survival8. According to the authors, chemotherapy-induced eyebrow loss is a psychological and emotional stressor that can adversely affect cancer patients, especially when combined with other stressors that cancer patients typically experience in their daily lives. Although eyebrow loss may appear trivial to the healthy population, anything that care providers can offer to their patients to provide relief from psychological and emotional stressors would be beneficial to those patients by helping them to better tolerate chemotherapy and achieve a better response to treatment, thereby producing a more positive outcome which may impact their survival.
Further investigation and testing may lead to other valuable applications of the EES — Essential Eyebrow Solution, including, but not restricted to, eyebrow thinning or loss due to thyroid disorders, ageing, and alopecia areata (non-cancer related).