Alexander Turkevych, Marta Turkevych, and Danylo Turkevych take an in-depth look at how treatments can affect collagen levels in the skin

MD, PhD, Assoc. Prof; Marta Turkevych, MD; Danylo Turkevych, MD
Aesthetic dermatology (AD) has roots dating back to antiquity. Aesthetic dermatology is based on principles no different from other medical specialties and can provide a serious adjunct to medical and surgical treatments. It is a rapidly developing part of dermatology in science and clinical practice. Concerns had been raised that AD might be a frivolous specialty, guided by a market that is aimed primarily at generating a profit. The focus, however, needs to be patient-centred and the work scientific to avoid any ethical dilemma. The acceptance of AD by the general population varies with traditions, cultural backgrounds and education. As AD has the potential of being preventive medicine at its best, the attitude toward AD may reflect the individual’s position to prevention, lifestyle, and quality-of-life (QOL)1–5.
Skin rejuvenation using procedures based on different injectables (HA, PDRN, PLLA, CaHA, PMMA), light sources (laser, IPL, LED), radiofrequency (monopolar, bipolar, fractional), and ultrasound, as well as combinations of these, have become popular in aesthetic medicine. Almost all treatment modalities based on these methods claim the main effect of their application to be the stimulation of neocollagenesis. This inspires the feeling that physiological neocollagenesis is a non-specific process, which can be stimulated through completely different ways.
Modulation of the dermal ECM via non-invasive or minimally-invasive aesthetic treatments, especially the stimulation of the collagen production de novo, should be arranged in a safe and physiological fashion. The ECM, with its insoluble scaffold — consisting mainly of stiff, mature fibrillar collagen — provides the structural support for the tissue and significantly determines its characteristic shape and dimensions. It is understood that collagen levels in the dermis continuously diminish with progressing age and can be further reduced as a result of photodamage or from the repeated initiation of remodelling processes.
The hypothesis that the injections of biological (PDRN, HA), chemical (PLLA), or mechanical/physical (CaHA, PMMA) substances or application of radiofrequency, ultrasound, or light sources can locally improve collagen levels in the dermis — consequently demonstrating some level of skin tightening and skin rejuvenation — appears to be reasonable. Indeed, the effect of neocollagenesis is generally considered to be long-term compared to the impact of other processes, such as hyaluronan and, consequently, water accumulation in a treated area. Such a long-term result is required in order to give credibility to the treatment methods that claim to improve the condition of the skin weeks and months after their application. This natural assumption contradicts, however, modern knowledge about the connective tissue structure and especially insight into its turnover, thus making the proclaimed results sometimes controversial and even theoretically impossible6–9.
Hyaluronic acid: what do we know?
Hyaluronan (HA) has high cross-species structural homology, which makes HA synthesized in bacteria or other species non-antigenic and non-immunogenic in humans. This property enables its widespread application for cosmetic uses. In fact, HA plays a central role in the dermal filler industry. By itself, it is the agent of choice for wrinkle fillers, preferred over collagen or other categories of smaller synthetic wrinkle fillers.
HA is a non-sulfated linear glycosaminoglycan polymer consisting of repeating disaccharide units of β-1,4 linked D-glucuronic acid (GlcUA) and β-1,3 linked N-acetyl-D-glucosamine (GlcNAc). HA is secreted to the extracellular matrix in most mammalian tissue. It is synthesized by three plasma membrane-bound hyaluronan synthases, HAS1, HAS2 and HAS3. During their synthesis, the nascent HA chains are extruded through pore-like structures into the extracellular space. Newly synthesized HA can be processed by hyaluronidase (HYALs) or broken down non-enzymatically by reactive oxygen species. Hyaluronidase hydrolyzes the hexosaminidic β (1–4) linkage between GlcNAc and GlcUA of the HA chain and releases small HA fragments. The half-life of HA varies in different organs. The turnover of HA is extremely high in circulation. In humans, the plasma half-life of HA is estimated to be about 2–6 min, resulting in a total turnover of 10–100 mg per day, the HA turnover in the whole body is estimated to take place within 3 days with a turnover of about 5 g per day. HA synthesis and degradation is also very dynamic at the cellular level. In cells, normal HA synthesis is activated transiently for cell division or motility, after which HA is rapidly cleared from the site by endocytic uptake and hyaluronidase-catalyzed hydrolysis10.
White adipocyte tissue (WAT) grows via cellular hyperplasia and volume expansion during development and a calorie surplus. However, the origin of white adipocytes and their developmental process, especially in adulthood, are complicated and remain to be completely understood; however, much progress has been made in recent years11. The prevailing hypothesis is that a perivascular population of cells, which are large in number and resemble mural cells (pericytes and vascular smooth muscle cells), give rise to new adipocytes under proadipogenic conditions, such as a high-fat diet. During maturation of preadipocytes, they progressively change their shape and accumulate lipid droplets, a process that needs to be coordinated with the remodelling of the extracellular matrix (ECM) to accommodate the expanding cellular volume and intercellular space12. So, adipogenesis is spatially and temporally regulated by the ECM.
Recent progress in genetics and the development of serum stable hyaluronidase enzymes have advanced our understanding of HA in the metabolism, especially the role of HA in adipogenesis and adipose tissue metabolism. Future studies should leverage the advancement in genetically engineered animal models, in which tissue specifically overexpresses an HA synthase or a hyaluronidase to carefully dissect the roles of HA in the adipose tissue and other metabolic organs10.
Caveolae, cavins and caveolins
Role in the ECM regulation and dermal adipose tissue reversible dedifferentiation
Skin ageing leads to various macroscopic and mesoscopic alterations within the structure of the individual skin layers as well as in the interactions between these layers, thereby affecting their mechanical stability and resulting in the appearance of the typical signs of ageing. These alterations affect both the composition of the skin cell population as well as the interactions between the skin cells and the extracellular matrix (ECM)13, 14.
Plasma membranes of eukaryotic cells have spatially heterogeneous structures containing lipid clusters enriched in cholesterol and sphingolipids, which are referred to as lipid rafts. Lipid rafts can appear in the form of planar structures or caveolae — plasma membrane invaginations forming nanodomains with a typical size of 50–100 nm, which are especially prevalent in mechanically stressed cells. They are involved in rapid adaptation to cellular volume changes, in various signal transduction processes, and in the processes of endo- and exo-cytosis.
Caveolae are enriched with a number of characteristic proteins, such as cavins and caveolins (Cav’s). The presence of caveolin-1 (Cav-1), which is the principal structural component of caveolae, is necessary for their characteristic appearance. Cav-1 is not exclusively localized to the plasma membrane, but is also found in different intracellular compartments, and can be transported in extracellular vesicles, providing a long-range mechanism of communication inside tissues or even between adjacent tissues.
Caveolin-1 (Cav-1) appears to be both a pathophysiological contributor and a target in different inflammatory and hyperproliferative skin conditions as well as in skin ageing. Skin fibroblasts demonstrate an up-regulation of Cav-1 expression both in chronological and UV-induced ageing, and such an up-regulation was observed both in vitro and in vivo.
Cav-1 expression
Typical alterations in ageing skin involve a reduction of the dermis thickness, a significant expansion of the dermal white adipose tissue as well as modifications to the distribution of hyaluronan, impairment of autophagic flux, a reduction of collagen expression and an increase in tissue inflammation. All of these phenomena can be connected to changes in Cav-1 expression in the ageing skin. Modified expression of Cav-1 can also significantly influence the mechanical properties of individual skin layers, thus changing the total mechanical stability of the layered composite skin/WAT, leading to structural modifications to the surface of the ageing skin. Selective reduction of Cav-1 expression has the potential to exert anti-ageing effects on skin changes under pathological conditions as well as over the course of the ageing process and can be affected by different external chemical and physical factors. Cav-1 demonstrates a stratified pattern of differential expression in various skin layers. High expression of Cav-1 is seen at the stratum granulosum/stratum corneum interphase and in the stratum basale as well as in the dermis, especially in association with connective tissue and endothelial cells.
“In the presence of HA, the majority of the TGF-β receptors are partitioned into Cav-1 lipid raft-associated pools, which significantly attenuates TGF-β1 signalling. As a result, collagen synthesis in response to TGF-β1 must be modulated by the presence of HA. “
Cav-1 expression correlates with ageing both in vitro and in vivo. It is strongly up-regulated in human diploid fibroblasts displaying a senescent phenotype in vitro. Overexpression of Cav-1 leads to a higher density of caveolae in the plasma membrane as well as morphological modifications of these cells, mainly concerning their focal adhesion and formation of actin stress fibres. Human corneal epithelial cells also demonstrate a continuous increase of Cav-1 levels with ageing, and in aged subjects, these cells display almost five times more caveolae than in young subjects15.
In the presence of HA, the majority of the TGF-β receptors are partitioned into Cav-1 lipid raft-associated pools, which significantly attenuates TGF-β1 signalling. As a result, collagen synthesis in response to TGF-β1 must be modulated by the presence of HA. It was proposed that the availability of exogenous HA leads to a co-localization of CD44 and TGF-β into Cav-1 lipid raft-associated pools. This effect was observed only with high molecular weight HA (HMW- HA) of 2 × 106 Da. In contrast, low molecular weight HA (LMW-HA) of 65,000 Da was unable to antagonize the effect of TGF-β1. This demonstrates how not only the content but also the sub-type of HA can influence the local collagen production in the tissue. Also, stimulation with TGF-β1 commonly enhances the total amount of HA in skin fibroblasts through an enhanced expression of hyaluronan synthase, whereby the molecular size of newly produced endogenous HA is dependent on the expression levels of a hyaluronan-binding protein involved in hyaluronan depolymerization (HYBID). In intact human skin, the content of TGF-β1 receptors positively correlates with HAS2 and negatively correlates with HYBID expression. This mechanism works only for HMW-HA, but not for depolymerized HA (as in the case of inflammation or aged skin)15–17.
Caveolae are flask-shaped plasma membrane specializations. A 22-kDa protein, caveolin, is a principal component of caveolar membranes in vivo. As recent evidence suggests that caveolae may participate in G‑protein-coupled signalling events, the potential interaction of caveolin with heterotrimeric G-proteins was investigated. Using cell fractionation techniques, it was discovered that mutational or pharmacologic activation of G‑proteins prevents its cofractionation with caveolin. In a second independent approach, the interaction of G‑proteins with caveolin was directly examined. For this purpose, caveolin as a glutathione S-transferase fusion protein was recombinantly expressed. Using an in vitro binding assay, it was found that caveolin interacts with G‑protein α subunits (Gs, Go, and Gi). Mutational or pharmacologic activation (with guanosine 5′-O-(thiotriphosphate)) of G‑proteins subunits prevents this interaction, indicating that the inactive GDP-bound form of G‑proteins subunits preferentially interacts with caveolin. This G‑protein binding activity is located within a 41-amino acid region of caveolin’s cytoplasmic N-terminal domain. Further functional analysis shows that a polypeptide derived from this region of caveolin effectively suppresses the basal activity of purified G proteins, apparently by inhibiting GDP/GTP exchange. This caveolin sequence is homologous to a region of the Rab GDP dissociation inhibitor, a known inhibitor of GDP/GTP exchange for Rab proteins. These data suggest that caveolin could function to negatively regulate the activation state of heterotrimeric G‑proteins18.
Fat cells
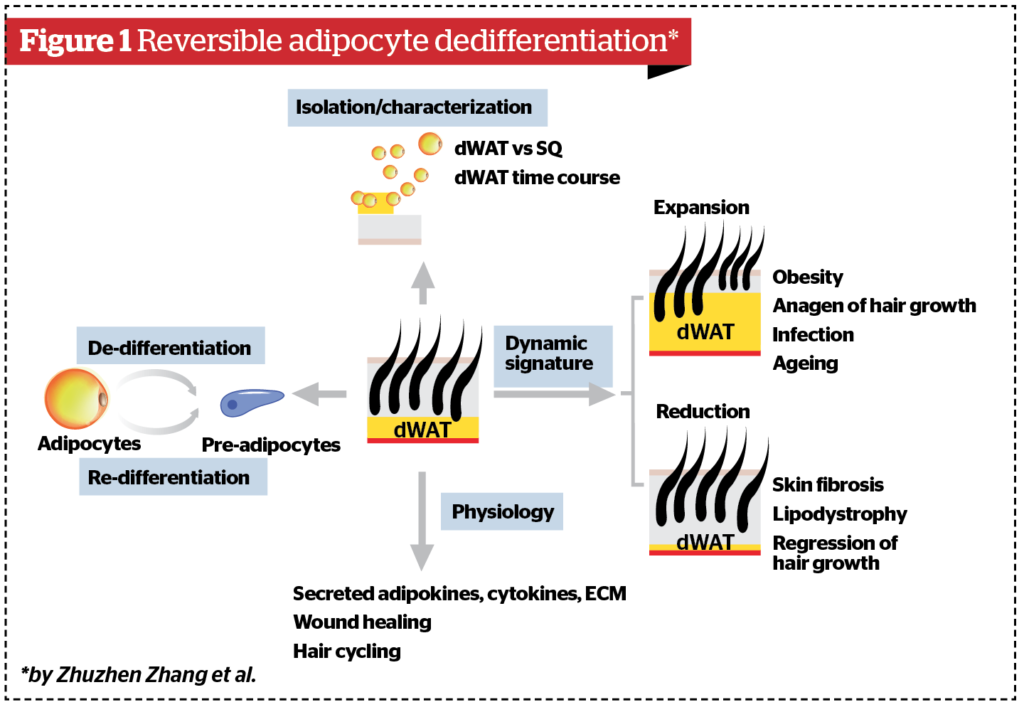
White adipose tissue evolved as the principal site for energy storage for vertebrate animals. White adipocytes, or ‘fat cells,’ are characterized by the presence of a single large lipid droplet (‘unilocular’ adipocytes). Their classical function is to serve as a storage compartment for excess energy; however, we now recognize that adipocytes are dynamic endocrine organs that secrete various hormones and cytokines (adipokines) to control nutrient homeostasis, energy balance, and immune cell regulation. White adipocytes arise through the process of adipocyte differentiation, or ‘adipogenesis’. In rodents and humans, most adipocytes arise pre- or perinatally, with fat cell numbers largely established by the adolescent period. In adulthood, there is relatively little fat cell turnover in the major, well-characterized, subcutaneous and intra-abdominal WAT deposits. In response to certain challenges (e.g. caloric excess, cold exposure, injury), de novo fat cell differentiation can occur, through the recruitment of resident adipocyte precursors (preadipocytes). However, physiological changes in subcutaneous or intra-abdominal WAT mass are largely driven by alterations in lipid content (lipogenesis and lipolysis). Upon various challenges, the de-differentiated cells proliferate and re-differentiate into adipocytes. In addition, manipulation of dWAT highlighted an important role for mature dermal adipocytes for hair cycling and wound healing. Altogether, these observations unravel a surprising plasticity of dermal adipocytes and provide an explanation for the dynamic changes in dWAT mass that occur under physiological and pathophysiological conditions, and highlight the important contributions of dWAT towards maintaining skin homeostasis19–22.
The model of dermal adipose tissue reversible dedifferentiation was described by Zhuzhen Zhang and is shown in Figure 1.
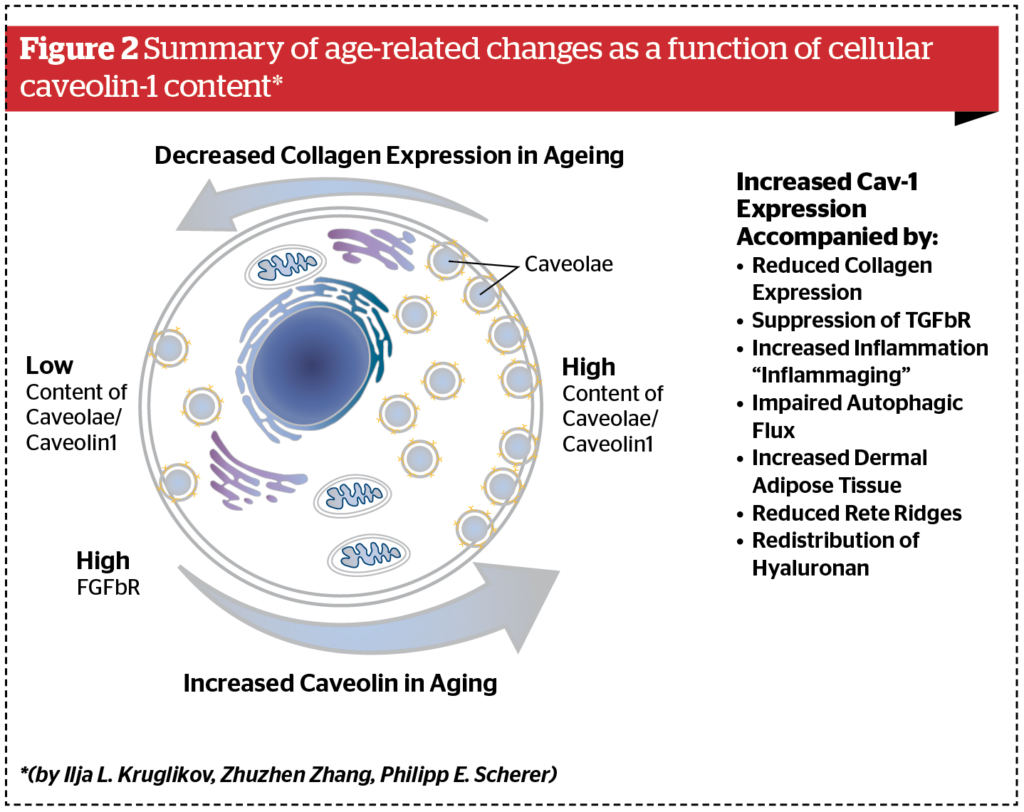
An upregulation of Cav-1 demonstrates a strong correlation with ageing phenotypes, both in chronological and UV-induced ageing in vitro and in vivo (Figure 2). The modification of Cav-1 content significantly affects collagen production and inflammation in the ageing process. The age-dependent modulation of Cav-1 also leads to modifications of the content and distribution of hyaluronan in the skin, and contributes to an impairment of the autophagic flux in ageing cells. Recent results reported in areas such as skin ageing, fibrosis and psoriasis suggest that the above-mentioned interactions are not merely correlations. Rather, Cav-1 is a pathophysiological factor and should be a target for different inflammatory and hyper-proliferative skin conditions, as well as in skin ageing. The modulation of Cav-1 expression can significantly influence the mechanical properties of single skin layers, as well as the adhesion between adjacent layers. This will affect the mechanical stability of the layered composite skin/WAT. As such, scientists propose that selective targeting of Cav‑1 expression should lead to relevant anti-ageing effects in the skin15.
Collagen-inducing treatments in AD
Every treatment modality that claims the modification of dermal collagen levels is the main effect of its application comes up against at least three problems. One is that the upregulation of multiple stages of collagen production is not necessarily an indication of increased levels of mature collagen in the skin. While procollagen can be easily overexpressed after the application of different physical modalities, it can also be easily degraded, which results in a very slow net remodelling of mature collagen. The second problem is connected with the type of induced collagenesis — physiological (with a regular collagen network structure) or pathological (scar-like, with typical hyalinised collagen regions). Strengthening of the natural structure of the collagen network in ECM would be the ideal solution, guaranteeing that physiological processes are retained, but this structure is not necessarily in equilibrium and can be reduced relatively rapidly. Pathological neocollagenesis (fibrosis) is connected to the application of higher intensities and results in the production of scar-like, intra-tissue structures. These structures can significantly improve the skin rigidity; they are, however, pathologic in their nature. The areas with hyalinised collagen, which is the typical hallmark of keloid scars, will not be refilled with fibroblasts, making later remodelling in this area difficult or even impossible. And the third problem is the dynamic nature of the ECM remodelling, with different characteristics of the dynamic processes involved. Especially important here is the very slow remodelling dynamic of mature collagen that cannot be significantly modified under quasi-physiological conditions, thus almost elevating the ‘wounding’ of ECM to the status of effective non-invasive treatment23.
The degradation of collagen is a very sophisticated process, which has both extracellular and intracellular pathways. Extracellular degradation is mediated by some MMPs and is specific to rapidly growing or damaged tissue. Under physiological conditions, collagen degradation is primarily connected to an internal pathway, which is often named collagen phagocytosis. This process is independent of MMP-1 activity, but can be significantly modulated by MT1-MMP. It has been shown that the same subjects can have a very different relationship with the synthesis of procollagen and the expression of MMPs in different regions of the body. The areas with low sun exposure (chronological skin ageing) in older people demonstrate significantly decreased production of procollagen I mRNA and the increased expression of MMPs, when compared to the same body areas in younger people. In the areas of strong sun exposure (photoageing), the procollagen I mRNA production was, however, on average 2.9 times higher and the MMPs were more strongly overexpressed than in the areas with pure chronological ageing in the same subjects. Both processes (chronological and photo-induced ageing) led to the same endpoint—total collagen deficiency23–25.
Pathological neo-collagenogenesis —
mechanical or physical stimulation of collagen production
The significant migration and production of new fibroblast does not usually take place under non- or pathophysiological conditions. What we typically describe as a pathophysiological condition for collagenogenesis is a ‘foreign-body reaction’ as a result of the presence of permanent or semi-permanent particles in the tissues, injected for so-called ‘new collagen stimulation’ and causing fibrosis at the end. Among them, CaHAP and PMMA are two substances that play a central role in the market. It’s necessary to mention that commercialized CaHAP’s come in two different forms — 8–12 mcm particle size, branded on the market as PEG HA filler with 1% CaHAP, and at least three brands with >25 mcm particle size, branded as 30% CaHA fillers from Germany and Israel. Calcium hydroxyapatite (CaHAP) has bibliographic evidence of its capability to stimulate collagen synthesis in fibroblasts.

We need to mention that the biosafety of the 8–12 mcm particle size 1% CaHA has been tested with at least three different methods: MTT test, NRU test, and Kenacid Blue assay. Moreover, any possible effect on the structure, morphology, and viability of cells has been evaluated. The results obtained show that the 8–12 mcm particle size 1% CaHA does not cause any cytotoxic effect and does not affect the correct structure and morphology of cell cultures. Non-cytotoxic concentrations of the product with 8–12 mcm particle size with 1% CaHAP has been used to treat human fibroblasts to evaluate any possible increase in the collagen production. Due to this, we can tell that there are two different types of CaHA in AD: scientifically-evident, small, non-cytotoxic concentrations of CaHA with small particles, and concentrations of CaHA with larger particles, which are likely cytotoxic for the surrounding tissue and human fibroblasts with a lack of scientific, marketing-free evidence. We have searched the medical and biological literature and found some interesting data: recently, a proliferation-suppressive effect of HAPs nanoparticles against various cells, also cancer among them was reported. This study was aimed at assessing the translational value of HAPs both as a bone-regenerating material and as an anti-tumour agent27,28. Inhibition of tumour growth, prevention of metastasis, and enhancement of the survival rate of tumour-bearing rabbits treated were demonstrated. Activated mitochondrial-dependent apoptosis in vivo was confirmed and was it was also observed that a stimulated immune response was involved in the induced anti-tumour effect. With the HAPs rising concentration, the percentage of early and late apoptotic cells increased from 8.99 ± 1.4% to 13.08 ± 1.6%. Meanwhile, it was found that genes expressed by different types of immune cells were also up-regulated in the HAPs group (TNF, DCSTAMP, MRC1, CLEC7A, TLR4, RLA-DR-ALPHA, and GZMK), revealing that HAPs might have stimulated the innate immune system and recruited numerous immune cells, including macrophages, dendritic cells, mononuclear cells, and lymphocyte cells, to the tumour microenvironment. In addition, genes involved in cellular calcium transport (ATP2A1, SLC8A1, TRDN, SRL, CACNB1, RYR1, CASQ2, CALB1, STC1, and CACNA2D1) were also expressed differentially, suggesting that intracellular calcium homeostasis might be disturbed by HAPs26,27. There is also data that compares the cytotoxicity of HAPs on tumour and healthy cells. Due to the degree of difference between the inhibition of human cancer cells and healthy cells from HAPs treatments. The cancer cells (MGC-803, Os-732, and Bel-7402) and healthy cells (L-02, MRC-5, and HaCaT) were treated with low (0.14 g L−1) and high (0.56 g L−1) concentrations of HAPs for 3 days. The data represents that the inhibition of cancer cells was from 54.3 +/- 1.5 to 88.0 +/- 3.5, but inhibition of the healthy cells was also up to 29.0 +/- 4.0 [28].
So, the more we know about the positive anti-tumour effect of HAPs, the more we understand that it should be studied in AD much more than it has in the past, taking into account particle size, shape and concentration. On the other hand, we have to understand that changing the viscosity, elasticity, plasticity or concentration of the injectable by the practitioner means that it is off-label and can’t be used safely in the majority of the civilized countries around the world.
Quasi-physiological neo-collagenogenesis
The net amount of procollagen I protein in the dermis of photo-aged skin, which demonstrated a higher synthesis of procollagen I mRNA, is much lower. This apparently paradoxical result, which is mainly connected with the different expression of MMPs, allows some authors24 the opportunity to write about the potentially different pathophysiologies of chronological and photo-induced skin ageing23. The significant migration and production of new fibroblasts does not normally take place under quasi-physiological conditions, such as RF, lasers and chemical substances, as well as topical (TCA) or injectables (PLLA) procedures. These phenomena can, however, be of significant importance in moderate to severe tissue damage, for example, in wound healing or in skin resurfacing procedures, where quick and strong repair is required. What becomes apparent is the significant difference between the processes involved in skin rejuvenation and in wound healing, which are often considered to be very similar. Under quasi-physiological conditions, the connective tissue remodelling is in a dynamic balance between the production and destruction of its matrix components. Each instance of overproduction of collagen or other ECM components will, in the short-term, initiate the feedback mechanism, causing the stimulation of their enzymatic degradation and thus undoing a large part of the stimulatory effect. For example, the 532 nm KPT and the 1064 nm Q-switched Nd:YAG laser at intensities of 1.5 J/cm2 can increase the expression of pro-collagens I and III mRNAs and simultaneously decrease the expression of MMP-1 and MMP-210, consequently suppressing the cleavage of collagen. At the same time, the ablative application of Er‑Yag or CO2 laser could significantly increase the induction of procollagen I (by up to 7.5 times the original value, 21 days after the procedure) and III mRNAs and MMP-1 mRNA (by up to 40,000 times, seven days after the procedure), which also correlates with the dynamical change of MMP-1 protein levels. Whereas the net amount of procollagen I protein was also increased, up to six months after CO2 laser treatment, its increase was much lower than that of procollagen I mRNA, clearly demonstrating the balancing effect. The qualitative difference between the dynamic processes in the dermis after the application of these two treatment modalities may be connected with much stronger amplitudes of dynamic changes after applying the CO2 laser. It has even been assumed that different laser applications can cause different mechanisms of collagen destruction.
Let us estimate how realistic the changes actually are to the skin’s appearance through the quasi-physiological remodelling of the mature collagen network. We will assume the physiological half-life time of the mature collagen in the dermis to be 15 years. The increase of the procollagen I protein in the dermis will be taken to be 2.4, as, for example, the maximum value measured after one skin rejuvenation treatment with photodynamic therapy recorded seven days after the procedure. Assuming the whole procollagen I protein will be utilised to replace the mature protein and that the procollagen upregulation is constant during the whole time after the treatment (which is surely wrong and will cause us to significantly overestimate the results), the proportion of the mature collagen network that will be replaced during the first seven days after the treatment can be calculated to be approximately 0.15% (the upper limit of the estimation). Realistically, this value has to be reduced at least twice. It is absolutely unrealistic that such a modification can significantly improve the skin appearance and explains why mentioned procedures need to be repeated to maintain the visible results23, 29–31.
Physiological neo-collagenogenesis via A2A receptors stimulation and CAV-1 regulation
Chronic inflammation and inflammatory cells and cytokine networks play a pivotal role in one of the leading theories of ageing – inflammaging. Several cytokines are significantly elevated, including early proinflammatory factors (tumour necrosis factor-a (TNFa), interleukin-1 (IL‑1), and IL-6), late proinflammatory factors (e.g., anti-inflammatory molecules (e.g., IL-10, IL-1 receptor antagonist, and transforming growth factor b). Also, one of the key facts is the role of adenosine— a purine nucleoside that is released from a variety of cells in response to several types of stress32. It has been suggested that adenosine regulates inflammation via interaction with one or more of its four known receptors (A1, A2A, A2B, and A3)33. Stimulation of adenosine A2 and A3 receptors has been shown to alter the cytokine network by decreasing inflammatory cytokine secretion by macrophages in vitro34–36. In animal models of acute and chronic inflammation, non-selective adenosine receptor antagonists reverse the anti-inflammatory effects of methotrexate (MTX), a ‘gold standard’ of therapy in acute inflammation37–39.

It has been shown by many authors40 that a particular extract of DNA from the gonadic tissue of wild male sturgeons possesses cell renewal effects with possible anti-ageing benefits for skin moisture, thickness, elasticity and a reduction in skin wrinkles. In addition to inhibiting the early and late inflammatory cytokine cascade, PDRN increased circulating levels of IL-10 and IL-10 expression in the tissues. Taking into account all mentioned before and the fact, that the majority of injectables in aesthetic dermatology should be injected in the superficial fat pads (sWAT) we propose another model for the possible interactions (Figure 3) to explain how and why collagen can be stimulated; moreover, why some methods induce physiological, while others quasi-physiological or even pathological collagenogenesis.
We want to appeal to all our practitioner colleagues with the following message: not all of the rejuvenation methods currently offered on the market have a sufficient evidence base, especially regarding their safety. This does not mean that they are certainly dangerous, but it does mean they should be further studied. Again, we urge colleagues to understand the fact that any self-made changes to the officially recommended way of administering of the substances (HA, PLA, CaHa) — for example, mixing with other products or dissolving— is the responsibility of the doctor, who in this situation, knowingly or unconsciously, violates the official recommendations and, therefore, in cases of complications, can find themselves isolated in terms of legislation and patient safety. And last, but not least, beauty and youth also have a price, and it is our job to ensure it is not too high.
Declaration of interest None
Figures 1–3 © Dr Turkevych redrawn by Prime Journal
REFERENCES
- Gadebusch Bondio, M. Medizinische Ästhetik. Kosmetik und plastische Chirurgie zwischen Antike und früher Neuzeit. München, 2005, Wilhelm Fink Verlag
- Sahan, A., & Tamer, F. Restoring facial symmetry through non-surgical cosmetic procedures after permanent facial paralysis: A case report. Acta Dermatovenerologica Alpina, Pannonica et Adriatica, 26, 41–42., 2017
- Wollina, U., & Goldman, A. Botulinum toxin a and/or soft tissue fillers for facial rehabilitation. Wiener Medizinische Wochenschrift, 167, 92–95., 2017
- Wiesing, U. Ethical aspects of aesthetic medicine. Hautarzt, 60, 409–413., 2009
- Wollina, U., Piotr Brzezinski Aesthetic dermatology: What’s new, what’s true? Dermatologic Therapy. 2018;e12623
- Ilja L. Kruglikov, Neocollagenesis in Non-Invasive Aesthetic Treatments, Journal of Cosmetics, Dermatological Sciences and Applications, 2013, 3, 1-5
- J. Varani, R. L. Warner, M. Gharaee-Kermani, S. H. Phan, S. Kang, J. H. Chung, Z. Q. Wang, S. C. Datta, G. J. Fisher and J. J. Voorhees, Vitamin A Antagonizes Decreased Cell Growth and Elevated Collagen-Degrading Matrix Metalloproteinases and Stimulates Collagen Accumulation in Naturally Aged Human Skin, Journal of Investigative Dermatology, Vol. 114, No. 3, 2000, pp. 480- 486
- S. E. G. Fligiel, J. Varani, S. C. Datta, S. Kang, G. J. Fisher and J. J. Voorhees, Collagen Degradation in Aged/Photodamaged Skin in Vivo and after Exposure to Matrix Metalloproteinase-1 in Vitro, Journal of Investigative Dermatology, Vol. 120, No. 5, 2003, pp. 842-848
- G. J. Fisher, J. Varani and J. J. Voorhees, Looking Older. Fibroblast Collapse and Therapeutic Applications, Archive of Dermatology, Vol. 144, No. 5, 2008, pp. 666-672
- Y. Zhu, et al., Hyaluronan in adipogenesis, adipose tissue physiology and systemic metabolism, Matrix Biol., 2017
- C. Hepler, L. Vishvanath, R.K. Gupta, Sorting out adipocyte precursors and their role in physiology and disease, Genes Dev. 31 (2) (2017) 127–140
- E.C. Mariman, P. Wang, Adipocyte extracellular matrix composition, dynamics and role in obesity, Cell. Mol. Life Sci. 67 (8) (2010) 1277–1292
- Ilja L. Kruglikov, Scherer PE General theory of skin reinforcement. PLoS One 12, e0182865, 2017
- Ilja L. Kruglikov, Scherer, P.E., 2018. Skin aging as a mechanical phenomenon: the main weak links. Nutr. Healthy Aging 4, 291–307
- Ilja L. Kruglikov, Zhuzhen Zhang, Philipp E. Scherer, Caveolin-1 in skin aging – From innocent bystander to major contributor, Ageing Research Reviews, Volume 55, November 2019
- Ito, T., Williams, J.D., Fraser, D.J., Phillips, A.O., 2004a. Hyaluronan regulates TGF-ß1 receptor compartmentalisation. J. Biol. Chem. 279, 25326–25332
- Nagaoka, A., Yoshida, H., Nakamura, S., Morikawa, T., Kawabata, K., Kobayashi, M., Sakai, S., Takahashi, Y., Okada, Y., Inoue, S., 2015. Regulation of hyaluronan (HA) metabolism mediated by HYBID (HYaluronan Binding Protein involved in HA Depolymerization, KIAA1199) and HA synthases in growth factor-stimulated fibroblasts. J. Biol. Chem. 290, 30910–30923
- 18. Shengwen Li, Takashi Okamoto, Miyoung Chun, Massimo Sargiacomo, James E. Casanova, Steen H. Hansen, Ikuo Nishimoto and Michael P. Lisanti, Evidence for a Regulated Interaction between Heterotrimeric G Proteins and Caveolin, J Biol Chem. 1995 Jun 30;270(26):15693-701
- Zhuzhen Zhang, Mengle Shao, Ilja Kruglikov, Rana K. Gupta, Philipp E. Scherer, Dermal adipose tissue has high plasticity and undergoes reversible dedifferentiation in mice, J Clin Invest. 2019
- Kershaw EE, Flier JS. Adipose tissue as an endocrine organ. J Clin Endocrinol Metab. 2004;89(6):2548-2556.
- Arner P, et al. Dynamics of human adipose lipid turnover in health and metabolic disease. Nature. 2011;478(7367):110-113.
- Strawford A, Antelo F, Christiansen M, Hellerstein MK. Adipose tissue triglyceride turnover, de novo lipogenesis, and cell proliferation in humans measured with 2H2O. Am J Physiol Endocrinol Metab. 2004;286(4): E577-588
- Ilja L. Kruglikov. Neocollagenesis in Non-Invasive Aesthetic Treatments, Journal of Cosmetics, Dermatological Sciences and Applications, 2013, 3, 1-5
- J. H. Chung, J. Y. Seo, H. R. Choi, M. K. Lee, C. S. Youn, G. Rhie, K. H. Cho, K. H. Kim, K. C. Park and H. C. Eun. Modulation of Skin Collagen Metabolism in Aged and Photoaged Human Skin in Vivo, Journal of Investigative Dermatology, Vol. 117, No. 5, 2001, pp. 1218-1224
- H. Lee, C. M. Overall, C. A. McCulloch and J. Sodek, A Critical Role for the Membrane-Type 1 Matrix Metalloproteinase in Collagen Phagocytosis, Molecular Biology of Cell, Vol. 17, No. 11, 2006, pp. 4812-4826
- Kun Zhang, Yong Zhou, Cong Xiao, Wanlu Zhao, Application of hydroxyapatite nanoparticles in tumor-associated bone segmental defect, Science Advances 02 Aug 2019: Vol. 5, no. 8
- Wei Tang, Yuan Yuan, et al., Differential cytotoxicity and particle action of hydroxyapatite nanoparticles in human cancer cells, NANOMEDICINE VOL. 9, NO. 3
- Yingchao Han, Shipu Li, Xianying Cao, Different Inhibitory Effect and Mechanism of Hydroxyapatite Nanoparticles on Normal Cells and Cancer Cells In Vitro and In Vivo , Sci Rep. 2014; 4: 7134
- J. S. Orringer, S. Kang, T. M. Johnson, D. J. Karimipour, T. Hamilton, C. Hammerberg, J. J. Voorhees and G. J. Fischer, Connective Tissue Remodeling Induced by Carbon Dioxide Laser Resurfacing of Photodamaged Human skin, Archives of Dermatology, Vol. 140, No. 11, 2004, pp. 1326-1332
- C. Spock, A. I. Metelitsa, J. Kaufman and J. B. Green, Lasers and Light Sources to Activate Fibroblasts, Cosmetical Dermatology, Vol. 25, No. 1, 2012, pp. 27-33
- J. S. Orringer, C. Hammerberg, T. Hamilton, T. M. Johnson, S. Kang, D. L. Sachs, G. J. Fisher and J. J. Voorhees, Molecular Effects of Photodynamic Therapy for Photoaging, Archives of Dermatology, Vol. 144, No. 10, 2008, pp. 1296-1302
- Jacobson KA, Gao ZG. Adenosine receptors as therapeutic targets. Nat Rev Drug Discov 2006;5:247–64
- Cronstein BN. Adenosine, an endogenous anti-inflammatory agent. J Appl Physiol 1994;76:5–13
- Gomez G, Sitkovsky MV. Targeting G protein-coupled A2a adenosine receptors to engineer inflammation in vivo. Int
J Biochem Cell Biol 2003;35:410–4 - Hasko G, Szabo C, Nemeth ZH, Kvetan V, Pastores SM, Vizi ES. Adenosine receptor agonists differentially regulate IL-10, TNF-a, and nitric oxide production in RAW 264.7 macrophages and in endotoxemic mice. J Immunol 1996;157: 4634–40
- Link AA, Kino T, Worth JA, McGuire JL, Crane ML, Chrousos GP, et al. Ligand-activation of the adenosine A2a receptors inhibits IL-12 production by human monocytes. J Immunol 2000; 164:436–42
- Asako H, Wolf RE, Granger DN. Leukocyte adherence in rat mesenteric venules: effects of adenosine and methotrexate. Gastroenterology 1993;104: 31–7
- Cronstein BN, Naime D, Ostad E. The antiinflammatory effects of methotrexate are mediated by adenosine. Adv Exp Med Biol 1994; 370: 411–6
- Montesinos MC, Yap JS, Desai A, Posadas I, McCrary CT, Cronstein BN. Reversal of the antiinflammatory effects of methotrexate by the nonselective adenosine receptor antagonists theophylline and caffeine: evidence that the antiinflammatory effects of methotrexate are mediated via multiple adenosin ae receptors in rat adjuvant arthritis. Arthritis Rheum 2000; 43: 656–63
- Alessandra Bitto, Francesca Polito et al., Polydeoxyribonucleotide Reduces Cytokine Production and the Severity of Collagen-Induced Arthritis by Stimulation of Adenosine A2A Receptor, Arthritis & Rheumatism
Vol. 63, No. 11, November 2011, pp 3364–3371