As hyperpigmentation continues to impact quality of life worldwide, emerging topicals such as cysteamine, 2-MNG and thiamidol offer promising, evidence-based alternatives to traditional therapies

Skin colour depends on the amount and distribution of melanin, a complex polymer and pigment produced by melanocytes1–3. Melanin is also a skin chromophore, contributing to colour perception by absorbing and reflecting light2. Cutaneous hyperpigmentation can result from increased melanin production, a higher density of active melanocytes, or deposition of exogenous substances1–3. Various internal and external factors can contribute to hyperpigmentation, which explains why treatment favours a multimodal approach that incorporates both preventative measures and active interventions.
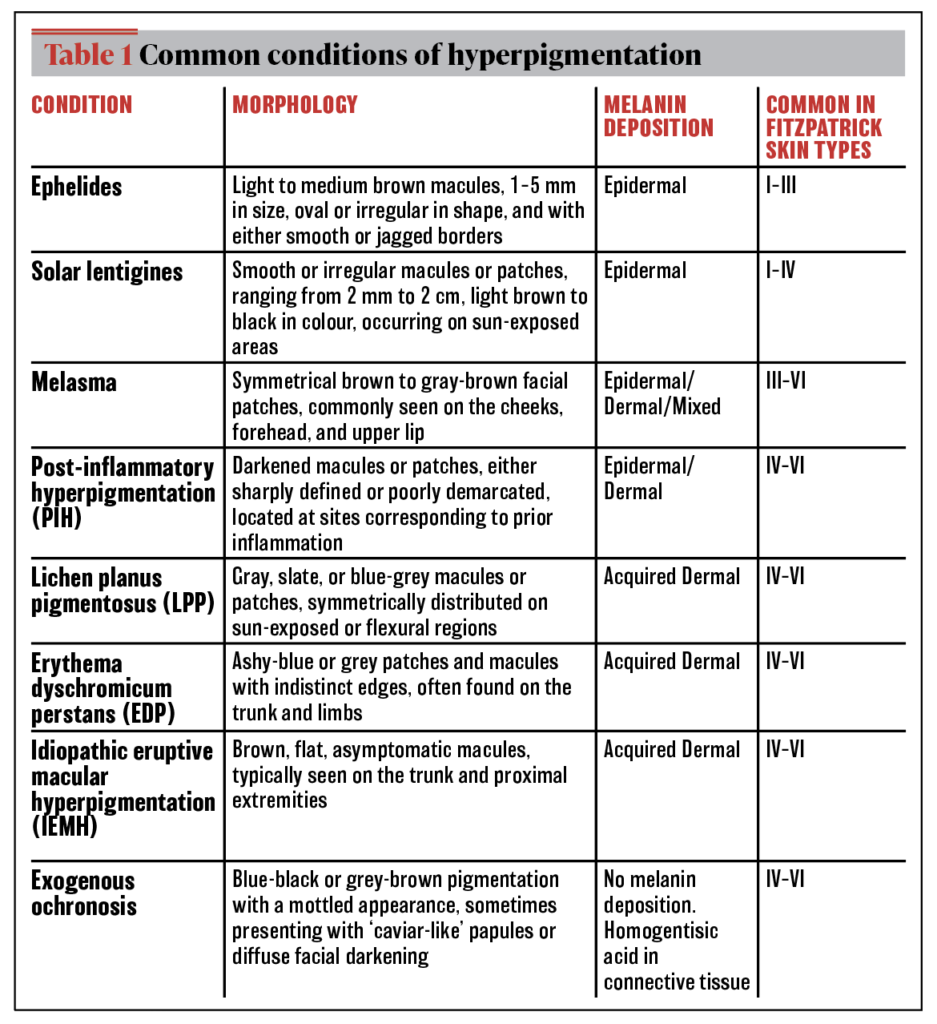
Hyperpigmentation varies in presentation and clinical context, underscoring its status as an umbrella term for various conditions (Table 1). While most conditions can be addressed within months of appropriate treatment, more refractory or resistant conditions may require biopsy for appropriate diagnosis. Pigmentary conditions are some of the most common chief ambulatory presenting complaints of dermatology patients, while also disproportionately affecting women and individuals of colour.
The melanogenesis pathway drives the development of therapeutic, enzymatic targets in hyperpigmentation. The enzyme tyrosinase is the rate-limiting enzyme that catalyses the synthesis of melanin from tyrosine4. This explains why tyrosinase inhibitors encompass the standard treatments for hyperpigmentation. Other potential targets include inhibiting melanocyte stimulation and melanosome transfer, antagonising various melanocyte cell receptors, and degrading melanin formed in keratinocytes4,5. Although hyperpigmentation is not considered physiologically harmful, there is a significant impact on quality of life with a considerable burden on mental health, negative self-perception, and stigma6,7. Facial pigmentary conditions are commonly associated with depression, isolation, anxiety, and hyper-self-awareness, negatively impacting an individual’s health-related quality of life6.
Treatment options include cosmeceuticals, prescription topicals, oral medications, and procedures such as laser and microneedling. Photoprotection remains one of the most important ways to prevent hyperpigmentation and sustain treatment efficacy. Benefits, risks, and side effects should be considered when selecting treatment options. For instance, corticosteroids may result in skin atrophy and hypopigmentation, while retinoids may also lead to photosensitivity, peeling, and xerosis. More severe sequelae, such as exogenous ochronosis, can result from hydroquinone misuse8. Generally, hyperpigmentation may require months or even years to achieve clinically significant results9,10. A prolonged treatment course may contribute to poor patient adherence and satisfaction. Ultimately, there is room for improvement in the treatment landscape for hyperpigmentation, calling for more timely, effective, and safer approaches.
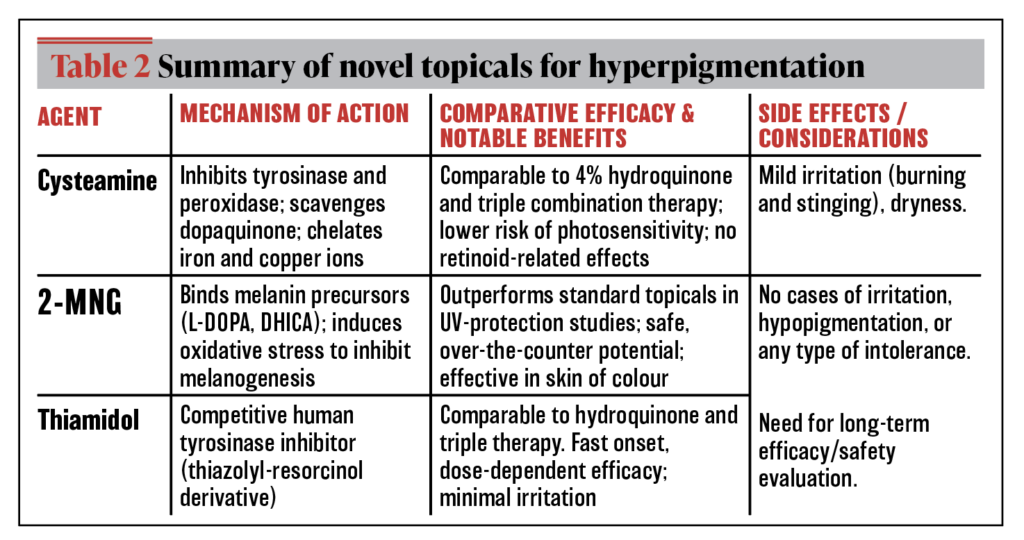
The industry has responded to the demand for novel treatments for hypermelanosis with the development of three new topicals: cysteamine, 2-MNG, and thiamidol (Table 2). We will review these agents, their pathogenesis, and therapeutic potential while exploring their potential to treat recalcitrant or dermal pigmentary conditions.
Cysteamine
The preclinical development of cysteamine started in the 1960s, with animal models and ex vivo work expanding into the 1980s and early 2000s11–14. These studies served as the biochemical basis for later clinical trials, with the first pivotal randomised controlled trial published in 201515. Since then, multiple clinical trials have demonstrated the value of cysteamine as an equally effective method to treat pigmentation compared to more established treatments.
Cysteamine ex vivo studies
Cysteamine is a compound initially identified as an aminothiol that results from the natural degradation of L-cysteine. Its thiol-based molecular structure makes it a known inhibitor of tyrosinase and peroxidase, two key enzymes in melanin synthesis12,16. The compounded function of thiols contributes to cysteamine’s overall depigmenting effects, as thiols may also scavenge dopaquinone, chelate iron, and copper ions16,17. Removing dopaquinone from the pathway will reduce melanin production, while chelating ions disrupt Fenton reactions that lead to pigment synthesis. Melanocytes were cultured with cysteamine, which decreased melanocyte hyperactivity and increased intracellular glutathione levels16,18. Glutathione has been shown to shift melanin synthesis away from eumelanin formation18.
Cysteamine clinical studies
A double-blind, randomised 2015 trial was the first to evaluate the efficacy of cysteamine on melasma15. After four months of nightly use, 5% cysteamine cream showed statistically significant reductions in MASI score and melanin content with colourimetry compared to placebo15. These preliminary results prompted investigators to use a more stabilised topical formulation and compare it with standard treatment regimens. Comparative studies began in 2020 with a pivotal Brazilian randomised controlled trial that compared 5% cysteamine to 4% hydroquinone among women with melasma19. Forty participants used either agent nightly on their hyperpigmented facial areas for 120 days19. All participants were instructed to wear tinted sunscreen daily19. Cysteamine had comparable therapeutic effects to hydroquinone, with similar scores on primary and secondary assessment measures. The strong depigmenting effects of cysteamine suggest it may be a feasible alternative for patients with melasma. Another mainstay of topical melasma treatment is Kligman’s Formula, or triple combination therapy (TTC), which is composed of hydroquinone, a retinoid, and a low-potency steroid. Even compared to the TTC, cysteamine demonstrated similar efficacy and a similar onset of action at 4 weeks20. Cysteamine’s potential for long-term daily use is important to consider, given that hydroquinone-based treatments must be cycled. The lack of retinoid in cysteamine also reduces the risk of photosensitivity. Other side effects, such as skin dryness, atrophy, irritation, and erythema, were less prevalent among the cysteamine group15,19–21. One study found that individuals applying cysteamine for melasma had a reduction in disease-related burden, but this difference was not statistically significant when comparing those taking cysteamine with those taking hydroquinone22.
Topical cysteamine has also been used for other common pigmentary conditions, such as acne-induced post-inflammatory hyperpigmentation (A-PIH) and solar lentigines21,23,24. In just 12 weeks, daily cysteamine application resulted in a statistically significant improvement in all objective measures of pigmentation in subjects with A-PIH and solar lentigines23,24. These statistically significant results also translated to high subject satisfaction23,24.
Opportunities for cysteamine research
Although cysteamine has become a viable option for treatment, there is still a need for more research. Current literature on cysteamine use only includes controlled trials for 12 or 16 weeks15,20-24. Long-term, observational studies are needed. The role of topical cysteamine in refractory pigmentation has only been studied with case reports. One report describes the treatment of biopsy-proven lichen planus pigmentosus-like drug reaction25. The individual applied 5% cysteamine cream daily for 6 months along with eight separate treatment sessions with 1,064 nm Q-switched Nd:YAG laser25. After the patient failed numerous conventional therapies, this combination treatment yielded significant improvement. The risk of exogenous ochronosis with long-term cysteamine use also needs to be further studied. Given cysteamine’s numerous therapeutic targets, there may be promise in the agent for treating conditions with dermal melanophages. Further studies are needed to establish evidence-based guidelines.
2-Mercaptonicotinoyl glycine
Another emerging agent in hyperpigmentation management includes 2-mercaptonicotinoyl glycine (2‑MNG), a new proprietary molecule.
2-MNG mechanism of action
Unlike conventional therapies that focus on inhibiting key melanogenesis enzymes, 2-MNG has a novel mechanism: it binds melanin precursors, i.e., L-DOPA, DHICA, and DHI, to prevent their integration to form melanin26,27. This unique pathogenesis ensures depigmentation without inducing melanocyte cell death. Instead, 2-MNG induces oxidative stress, reducing the chemical reactivity across the melanocyte environment and making them more vulnerable to reactive oxygen species (ROS)26,28. Epidermal melanocytes are particularly susceptible to oxidative stress, serving as an ideal target to reduce melanogenesis28.
2-MNG in vitro and ex vivo studies
Investigators used an in vitro technique called high-throughput screening (HTS) to blindly screen a large library of molecules that could potentially inhibit melanin production in human melanocytes26. Safety, environmental impact, and skin penetration were also considered26. From this collection of molecules, thiopyridone was isolated as one of the more potent chemicals for depigmentation26. This approach resulted in 192 new formulations that were tested using human skin explants and reconstructed epidermis models (ex vivo), leading to the discovery of 2-MNG26. 2-MNG’s potency may be attributed to its structural similarity with thiouracil (TU), which also interacts with melanin intermediates26. The multi-step study of the biological assay confirmed that 2-MNG, the newest member of the thiopyridine family, can be used to treat hyperpigmentation.
2-MNG clinical studies
The first series of preclinical studies on 2-MNG were designed to evaluate its ability to prevent pigmentation after controlled ultraviolet violet (UV) light exposure. Studies were conducted in Mauritius, Shanghai, Paris, and Budapest27,29. Controlled UV light was administered on the backs of subjects in designated areas where 2-MNG formulations and other conventional serums were administered27,29. Although the duration, frequency, and strength of light exposure varied across studies, they all supported the photoprotective and depigmenting role of 2-MNG27,29. Specifically, 2-MNG prevented darkening from day 12 through day 47, with a stronger and longer-lasting effect27. 2-MNG formulated with other topicals, such as a salicylic acid derivative, and a broad ultraviolet-A (UVA) absorber had the earliest and strongest effect, starting on day 927. Regardless of whether the topical was solely 2-MNG or a combination formulation, formulas with 2-MNG outperformed all standards and controls26, 29.
2-MNG has been studied for the use of acne-induced post-inflammatory hyperpigmentation, with a sample population of individuals from Mauritius with predominantly darker-pigmented skin (Fitzpatrick scale IV‑VI)30,32. Subjects (age 14-46) used 2-MNG as a prospective agent for acne-induced PIH30. Results showed a significant improvement in the global post-acne hyperpigmentation index (PAHPI) score and notable improvements in the mean darkness of lesions among the 2-MNG group30. Pigmentation intensity and lesion size also showed statistically significant reductions30. Subjects also acknowledged benefits, with results showing a moderate improvement in self-perceived PAHPI and a significant decrease in global stigmatisation by the end of the study. Subject satisfaction was also high, with most individuals noting that 2-MNG was more effective than other topical hyperpigmentation treatments previously trialled30. Continuous work on optimising formulation is needed, as 2-MNG offers an over-the-counter, potentially safer alternative for treating pigmentation.
Thiamidol
Thiamidol is another topical cosmeceutical product developed after ten years of laboratory research. Although it has been in use since 2019, momentum towards its use has increased in recent years.
Thiamidol in vitro and ex vivo studies
While topical cysteamine and 2-MNG have distinct mechanisms of inhibiting melanogenesis, thiamidol focuses on inhibiting tyrosinase. Similar to 2-MNG, researchers utilised an active screening approach to search for agents with the potential to inhibit melanin formation31. Investigators used recombinant human tyrosinase to screen a collection of over 50,000 compounds31. In vitro HTS was done to identify structural motifs that could inhibit human tyrosinase31. The newly discovered molecules were then compared with other well-known depigmenting agents (i.e., hydroquinone, kojic acid, and arbutin)31. From this extensive search, derivatives of thiazolyl-resorcinol showed the most promising effect on tyrosinase inhibition31. Thiamidol was the most potent inhibitor, with results demonstrating almost complete enzyme inhibition occurring at concentrations above 10 µmol/L31. Thiamidol’s most unique property is demonstrated by its strict competitive inhibition of human tyrosinase31,32. Thiamidol is better at inhibiting the human form of tyrosinase, compared to the other agents
that demonstrate greater inhibition of mushroom tyrosinase31,32. Using a three-dimensional model for human skin (ex vivo), purified human tyrosinase was used to compare the effect of arbutin, kojic acid, rhododendrol, hydroquinone, 4-butylresorcinol, and thiamidol31. Thiamidol was the most potent inhibitor of melanin production in these human skin models31.
Thiamidol clinical studies
Thiamidol also has clinical efficacy, with multiple studies demonstrating a statistically and clinically significant reduction in hyperpigmentation.
The first studies evaluating the effect of thiamidol on facial hyperpigmentation were published in 2019. A randomised controlled split-face study showed that using 0.2% thiamidol twice daily significantly reduced mild-to-moderate melasma severity as early as 2 weeks, with continued improvement over 12 weeks33. The compound was also well tolerated with minimal adverse effects over the 12-week study period33. Another study even went a step further to test the clinical efficacy of thiamidol four times per day, demonstrating a dose-dependent response34. Applying thiamidol four times per day was significantly more effective than twice a day application34. Subjects also found the formulation tolerable and suitable for incorporation into their daily routine34,35. Overall, this new agent has consistently demonstrated rapid, safe, and tolerable depigmenting effects across skin types and severity35.
Thiamidol has also been compared to other conventional treatments, such as hydroquinone and Kligman’s formula (varying strengths of hydroquinone, retinoic acid, and low-potency steroid)36,37. When comparing validated scales for melasma severity, i.e., MASI or mMASI, scores were equally comparable among the thiamidol combination group, hydroquinone, and triple combination topical therapy36,37. This makes it a promising alternative for individuals who may want to mitigate the possible cutaneous side effects of retinoids and hydroquinone. Other studies also evaluated the combination of thiamidol with other cosmeceuticals, such as hyaluronic acid38. Among individuals from Thailand, this study supported a synergistic benefit of combining thiamidol with hydrating agents such as hyaluronic acid38. Although the generalizability of this study may be limited due to the lack of ethnoracial diversity in the study population, this population is more susceptible to pigmentary disorders. Thus, incorporating thiamidol into a multimodal approach may be helpful for individuals who are more prone to resistant hyperpigmentation. Considering the relapsing and remitting course of melasma, further studies are needed to evaluate if the long-term use of thiamidol is safe and effective.
Recalcitrant pigmentation
Recalcitrant pigmentation persists or recurs despite appropriate therapy. Although aetiology and pathogenesis may differ among distinct types of resistant hyperpigmentation, several key features make them more difficult to treat: chronicity, deep pigment deposition in the dermal layer of the skin, and underlying triggers that are not being adequately addressed9,10,39,40. Certain conditions that are more difficult to treat include drug-induced pigmentation from drugs such as minocycline, amiodarone, antimalarials, or chemotherapy agents[1]. Prolonged use of these medications could lead to the presence of melanin and melanophages in the dermis. Other conditions include lichen planus pigmentosus, pigmented contact dermatitis, erythema dyschromicum perstans, exogenous ochronosis, maturational hyperpigmentation, periorbital hyperpigmentation, and nevus of Ota. Biopsies of these conditions can offer insight into why pigmentation is persistent or refractory, such as the presence of melanin-laden macrophages in papillary and/or reticular dermis, basal layer vacuolar degeneration or interface dermatitis, dermal fibrosis, or subtle signs of ongoing low-grade inflammation41–43. Once the diagnosis is confirmed, treatment options must also expand as patients may fail traditional options. There is limited literature on the role of cysteamine, 2-MNG, and thiamidol on refractory pigmentation, with only case reports demonstrating the efficacy of topical cysteamine 5% for drug-induced lichen-planus pigmentosus and resistant post-inflammatory hyperpigmentation25,40. Thiamidol and 2-MNG studies have only focused on the most common conditions, such as epidermal melasma, PIH, and solar lentigines24,27,30,33,34,37. Thus, there is an important need to explore how these new topicals can expand treatment options.
Conclusion
Treating pigmentation requires a comprehensive, multimodal approach. New treatment options have emerged, as novel compounds and molecular structures are screened and tested for their ability to prevent and treat pigmentation. As one of the most common patient complaints, it is increasingly important that dermatologists are aware of newer options. With additional research focused on efficacy, tolerability, and safety, these three new agents have the potential to become part of the gold standard for treating hyperpigmentation disorders.
- Declaration of interest Dr. Valerie Callender is a consultant for L’Oréal and Beiersdorf and has conducted clinical research with L’Oréal.
References
- Bolognia JL. Dermatology – E-Book: 2-Volume Set. 5th ed. Elsevier; 2024.
- Zamudio Díaz DF, Busch L, Kröger M, et al. Significance of melanin distribution in the epidermis for the protective effect against UV light. Sci Rep. 2024;14(1):3488. doi:10.1038/s41598-024-53941-0
- Lambert MW, Maddukuri S, Karanfilian KM, Elias ML, Lambert WC. The physiology of melanin deposition in health and disease. Clin Dermatol. 2019;37(5):402-417. doi:10.1016/j.clindermatol.2019.07.013
- Hida T, Kamiya T, Kawakami A, et al. Elucidation of Melanogenesis Cascade for Identifying Pathophysiology and Therapeutic Approach of Pigmentary Disorders and Melanoma. Int J Mol Sci. 2020;21(17):6129. doi:10.3390/ijms21176129
- Carlson JA, Linette GP, Aplin A, Ng B, Slominski A. Melanocyte receptors: clinical implications and therapeutic relevance. Dermatol Clin. 2007;25(4):541-557, viii-ix. doi:10.1016/j.det.2007.06.005
- Yadav A, Garg T, Mandal AK, Chander R. Quality of life in patients with acquired pigmentation: An observational study. J Cosmet Dermatol. 2018;17(6):1293-1294. doi:10.1111/jocd.12686
- Taylor A, Pawaskar M, Taylor SL, Balkrishnan R, Feldman SR. Prevalence of pigmentary disorders and their impact on quality of life: a prospective cohort study. J Cosmet Dermatol. 2008;7(3):164-168. doi:10.1111/j.1473-2165.2008.00384.x
- Lazar M, De La Garza H, Vashi NA. Exogenous Ochronosis: Characterising a Rare Disorder in Skin of Color. J Clin Med. 2023;12(13):4341. doi:10.3390/jcm12134341
- Ko D, Wang RF, Ozog D, Lim HW, Mohammad TF. Disorders of hyperpigmentation. Part II. Review of management and treatment options for hyperpigmentation. J Am Acad Dermatol. 2023;88(2):291-320. doi:10.1016/j.jaad.2021.12.065
- Pandya AG, Guevara IL. DISORDERS OF HYPERPIGMENTATION. Dermatologic Clinics. 2000;18(1):91-98. doi:10.1016/S0733-8635(05)70150-9
- Chavin W, Schlesinger W. Some potent melanin depigmentary agents in the black goldfish. Naturwissenschaften. 1966;53(16):413-414. doi:10.1007/BF00625789
- Atallah C, Charcosset C, Greige-Gerges H. Challenges for cysteamine stabilisation, quantification, and biological effects improvement. J Pharm Anal. 2020;10(6):499-516. doi:10.1016/j.jpha.2020.03.007
- Qiu L, Zhang M, Sturm RA, et al. Inhibition of melanin synthesis by cystamine in human melanoma cells. J Invest Dermatol. 2000;114(1):21-27. doi:10.1046/j.1523-1747.2000.00826.x
- Ito Y, Jimbow K, Ito S. Depigmentation of black guinea pig skin by topical application of cysteaminylphenol, cysteinylphenol, and related compounds. J Invest Dermatol. 1987;88(1):77-82. doi:10.1111/1523-1747.ep12465072
- Mansouri P, Farshi S, Hashemi Z, Kasraee B. Evaluation of the efficacy of cysteamine 5% cream in the treatment of epidermal melasma: a randomised double-blind placebo-controlled trial. Br J Dermatol. 2015;173(1):209-217. doi:10.1111/bjd.13424
- Besouw M, Masereeuw R, van den Heuvel L, Levtchenko E. Cysteamine: an old drug with new potential. Drug Discov Today. 2013;18(15-16):785-792. doi:10.1016/j.drudis.2013.02.003
- Kasraee B. Peroxidase-Mediated Mechanisms Are Involved in the Melanocytotoxic and Melanogenesis-Inhibiting Effects of Chemical Agents. Dermatology. 2002;205(4):329-339. doi:10.1159/000066439
- Smit NP, Van der Meulen H, Koerten HK, et al. Melanogenesis in cultured melanocytes can be substantially influenced by L-tyrosine and L-cysteine. J Invest Dermatol. 1997;109(6):796-800. doi:10.1111/1523-1747.ep12340980
- Lima PB, Dias JAF, Cassiano D, et al. A comparative study of topical 5% cysteamine versus 4% hydroquinone in the treatment of facial melasma in women. Int J Dermatol. 2020;59(12):1531-1536. doi:10.1111/ijd.15146
- Sachdev M, Grimes PE, Callender V, et al. Cysteamine Isobionic-Amide Complex Versus Kligman’s Formula for the Treatment of Melasma: Equal Efficacy and Rapid Onset of Action. J Drugs Dermatol. 2024;23(2):9-16. doi:10.36849/JDD.7428
- Desai S, Hartman C, Grimes P, Shah S. Topical Stabilized Cysteamine as a New Treatment for Hyperpigmentation Disorders: Melasma, Post-Inflammatory Hyperpigmentation, and Lentigines. J Drugs Dermatol. 2021;20(12):1276-1279. doi:10.36849/jdd.6367
- Sepaskhah M, Karimi F, Bagheri Z, Kasraee B. Comparison of the efficacy of cysteamine 5% cream and hydroquinone 4%/ascorbic acid 3% combination cream in the treatment of epidermal melasma. J Cosmet Dermatol. 2022;21(7):2871-2878. doi:10.1111/jocd.15048
- Ahmadi K, Miri A, Bizaval Z, et al. Assessing the Effectiveness of Stabilized Cysteamine 5% Cream Compared to Hydroquinone 4%/Ascorbic Acid 3% Combination Cream in Treating Acne-induced Post-inflammatory Hyperpigmentation: A Randomised, Controlled Study. J Clin Aesthet Dermatol. 2024;17(4):37-41.
- Saki N, Modabber V, Kasraei H, Kasraee B. Successful treatment of solar lentigines by topical application of stabilised cysteamine: A vehicle-controlled, double-blind randomised study. Health Sci Rep. 2024;7(2):e1930. doi:10.1002/hsr2.1930
- Murray TN, Rajanala S, Friedman PM. Improvement of Lichen Planus Pigmentosus-Like Drug Reaction Using a Combination of the Fractionated 1,550-nm Erbium-Doped Fiber Laser and Topical Cysteamine Cream. Dermatol Surg. 2023;49(11):1046-1048. doi:10.1097/DSS.0000000000003902
- Sextius P, Warrick E, Prévot-Guéguiniat A, et al. 2-Mercaptonicotinoyl glycine, a new potent melanogenesis inhibitor, exhibits a unique mode of action while preserving melanocyte integrity. Pigment Cell Melanoma Res. 2024;37(4):462-479. doi:10.1111/pcmr.13168
- de Dormael R, Sextius P, Bourokba N, et al. 2-Mercaptonicotinoyl glycine prevents UV-induced skin darkening and delayed tanning in healthy subjects: A randomised controlled clinical study. J Cosmet Dermatol. 2024;23(5):1745-1752. doi:10.1111/jocd.16200
- Denat L, Kadekaro AL, Marrot L, Leachman SA, Abdel-Malek ZA. Melanocytes as instigators and victims of oxidative stress. J Invest Dermatol. 2014;134(6):1512-1518. doi:10.1038/jid.2014.65
- Muller B, Flament F, Jouni H, et al. A Bayesian network meta-analysis of 14 molecules inhibiting UV daylight-induced pigmentation. J Eur Acad Dermatol Venereol. 2024;38(8):1566-1574. doi:10.1111/jdv.19910
- Demessant-Flavigny AL, Petkar G, Jodun D, Le Dantec G, Le Floc’h C, Kerob D. Efficacy of a 2-MNG-Containing Depigmenting Serum in the Treatment of Post-Inflammatory Hyperpigmentation. J Cosmet Dermatol. 2025;24(2):e16735. doi:10.1111/jocd.16735
- Mann T, Gerwat W, Batzer J, et al. Inhibition of Human Tyrosinase Requires Molecular Motifs Distinctively Different from Mushroom Tyrosinase. J Invest Dermatol. 2018;138(7):1601-1608. doi:10.1016/j.jid.2018.01.019
- T. Mann, V. Welge, J. Weise, D. Roggenkamp, L. Kolbe. Efficacy of Thiamidol, Niacinamide, Tranexamic acid, Cysteamine, Azelaic acid on melanin production in vitro. Virtual Poster Presentation presented at: 29th EADV Congress 2020; October 29, 2020; Virtual.
- Arrowitz C, Schoelermann AM, Mann T, Jiang LI, Weber T, Kolbe L. Effective Tyrosinase Inhibition by Thiamidol Results in Significant Improvement of Mild to Moderate Melasma. J Invest Dermatol. 2019;139(8):1691-1698.e6. doi:10.1016/j.jid.2019.02.013
- Philipp-Dormston WG, Vila Echagüe A, Pérez Damonte SH, et al. Thiamidol containing treatment regimens in facial hyperpigmentation: An international multi-centre approach consisting of a double-blind, controlled, split-face study and of an open-label, real-world study. Int J Cosmet Sci. 2020;42(4):377-387. doi:10.1111/ics.12626
- Frey C, Grimes P, Callender VD, et al. Thiamidol: A Breakthrough Innovation in the Treatment of Hyperpigmentation. J Drugs Dermatol. 2025;24(6):608-616. doi:10.36849/JDD.9093
- Bertold C, Fontas E, Singh T, et al. Efficacy and safety of a novel triple combination cream compared to Kligman’s trio for melasma: A 24-week double-blind prospective randomised controlled trial. J Eur Acad Dermatol Venereol. 2023;37(12):2601-2607. doi:10.1111/jdv.19455
- Lima PB, Dias J a. F, Cassiano DP, et al. Efficacy and safety of topical isobutylamido thiazolyl resorcinol (Thiamidol) vs. 4% hydroquinone cream for facial melasma: an evaluator-blinded, randomised controlled trial. J Eur Acad Dermatol Venereol. 2021;35(9):1881-1887. doi:10.1111/jdv.17344
- Disphanurat W, Srisantithum B. Efficacy and safety of 0.15% isobutylamido thiazolyl resorcinol combined with hyaluronic acid vs 0.15% isobutylamido thiazolyl resorcinol or hyaluronic acid alone in melasma treatment: A randomised evaluator-blind trial. J Cosmet Dermatol. 2021;20(11):3563-3572. doi:10.1111/jocd.14031
- Wang RF, Ko D, Friedman BJ, Lim HW, Mohammad TF. Disorders of hyperpigmentation. Part I. Pathogenesis and clinical features of common pigmentary disorders. J Am Acad Dermatol. 2023;88(2):271-288. doi:10.1016/j.jaad.2022.01.051
- Mathe N, Balogun M, Yoo J. A case report on the use of topical cysteamine 5% cream in the management of refractory postinflammatory hyperpigmentation (PIH) resistant to triple combination cream (hydroquinone, topical corticosteroids, and retinoids). J Cosmet Dermatol. 2021;20(1):204-206. doi:10.1111/jocd.13755
- Phansuk K, Vachiramon V, Jurairattanaporn N, Chanprapaph K, Rattananukrom T. Dermal Pathology in Melasma: An Update Review. Clin Cosmet Investig Dermatol. 2022;15:11-19. doi:10.2147/CCID.S343332
- Brown TM, Krishnamurthy K. Histology, Dermis. In: StatPearls. StatPearls Publishing; 2022. Accessed September 3, 2022. http://www.ncbi.nlm.nih.gov/books/NBK535346/
- Wang L, Xu AE. Four views of Riehl’s melanosis: clinical appearance, dermoscopy, confocal microscopy and histopathology. J Eur Acad Dermatol Venereol. 2014;28(9):1199-1206. doi:10.1111/jdv.12264





