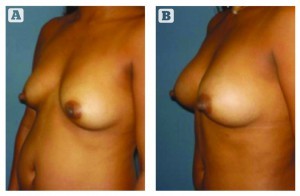
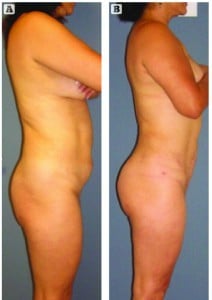
Autologous fat grafting meets all of the fundamental criteria of the ‘ideal’ augmentation: availability, minimal donor morbidity, and reproducible and predictable results. The standardisation of consistent harvesting, graft manipulations, use of platelet-rich plasma (PRP), preparation of the recipient area and transfer protocols are improving the ability to accurately predict volume enhancements, and realise long-term survival of the grafted tissues.
Autologous fat grafting offers a viable and safe alternative to soft tissue augmentation in select patients. Long-lasting, natural contouring and volume increases are attainable.
The transplantation of fat from one area of the body to another is a safe and effective procedure when performed by the hands of a qualified surgeon. However, it requires more than just the action of filling an area with fat tissue; successful fat transfer is also dependent on the regenerative action of adult stromal stem cells for survival.
For autologous fat grafting to emerge as a mainstream technique, it must be safe, yield reproducible results, and be based on stringent surgical principles. What began as an apparently simple technique of suctioning fat with implantation into areas for cosmetic and reconstructive purposes has now evolved into a complex menu of clinical choices. The four variables commonly considered important to the overall success of fat grafting include harvesting, handling, preparation of the recipient site, and transplantation or placement. These steps have developed and matured, leading to a multitude of techniques, technologies, and a range of opinions on each.
As a subset of the fat grafting process, we can now greatly enhance the quality of the fat with a variety of additives, including adipocyte-derived stem cells and platelet-rich plasma (PRP). Autologous fat will vascularise without reinjection if delivered in small aliquots. There is no cost for the fat and it is available in sufficient quantities from almost any patient.
History
Fat grafting is definitely not a new procedure. In 1893 Neuber first began to use small pieces of fat from the upper arm to reconstruct a depressed area of the face secondary to tuberculous osteitis, shortly followed by Czerny who, in 1895, used the fat from a large lipoma to fill a defect in the breast. These discoveries developed over the next century, with Bircoll1,2 reporting on the use of autologous fat from liposuction for contouring and filling defects. Asken3 then found that 90% of fat extracted by liposuction appears viable, assuming it is not traumatised either by handling or high suction pressure, and in 1989 Markman4 found that after lipografting, the number of fat cells may increase through differentiation of existing preadipocytes.
Illouz provided a number of important concepts, such as5:
- The cells live longer if mixed with normal saline and are kept at a moderate temperature
- They do not tolerate excessive manipulation, or major trauma.
Hudson6 then discussed the best donor sites and demonstrated greater cell size and lipogenic activity (lipoprotein lipase; LPL) in the gluteal femoral area compared with the abdomen.
Then, in 1995 Sidney Coleman demonstrated the long-term survival of liposuctioned fat transplanted into the nasolabial folds and that by placing the grafts in multiple tunnels, better nutritional support is possible the closer the graft is to normal tissue7. Coleman delineated a standardised reproducible process of fat harvesting, centrifugation, and injection to the face. That ‘structural fat grafting’ could be reliably replicated and a reproducible technique became clearer8.
As with early grafting to the face, fat grafting to the buttocks and breasts was also initially described in the late 1980s. Larger volume applications, especially to the breast, were met with more criticism, and were placed on hold for the next 20 years as a result of concerns regarding patient safety and lack of efficacy.
Roger Amar developed the FAMI technique of fat grafting in 2000, and in 2009 Giorgio Fischer presented a new technique of lipografting, known as the ‘Rice Technique’9.
Autologous fat grafting
Autologous fat grafting meets all of the fundamental criteria for the ‘ideal’ augmentation material: availability, minimal donor morbidity, reproducible and predictable results, while avoiding non-graft disease transmission or incompatibility. Considering these facts, autologous fat transfer provides a very appealing resource for soft tissue volume augmentation, with both small and large volumes. It is important to consider and follow the Guidelines for Fat Grafting10 (Table 1).
Fat transfer is absolutely the future of aesthetic medicine and cosmetic surgery, but it is not just about the fat cells — it is also about the fat tissue. It’s about improving the recipient bed and identifying what processes to minimise and/or optimise; understanding the stromal vascular elements and inserting the adult stem cells into the fat tissue, looking at specific reactions and promoting best practice; and understanding that excellent results are possible by working the physiology without any external manipulations of the fat tissue.
What are the best donor areas for fat transfer?
It is now known that fat tissue containing more α2 receptors (antilipolytic–adrenergic α2 receptors) and large amounts of LPL is the best for transfer, and that the tissue in the lower abdomen and pubis are the best source areas. LPL is an important enzyme for lipogenesis, as it can increase the size of the fat cells, which will also contain larger numbers of α2 receptors. Women generally produce more LPL in the subcutaneous fat tissue of the lower abdomen and gluteal femoral area.
Autologous fat grafting has proven metabolically more stable, and has more lipoprotein lipase activity and a greater percentage of α2 receptors11. When using fat for lipografting, areas with a large amount of fat tissue or zones with localised obesity are typically selected. These areas may cause problems at the recipient site. In the regions with localised obesity, the physiology of the fat tissue may vary. According to the studies of Danielle Lacasa12, there is an alteration in the viability of the fat cells in these areas: there is more fibrosis, less expression of adiponectin and leptin, and an increase in the proinflammatory cytokines. There is also an alteration in the normal lipolysis, with decreased β-adrenergic action in fat tissue with fibrosis. It is easy to understand that the quality of the fat tissue varies and that some fat used for grafting may give different results.