Safety assessments
Patient safety was estimated by the severity of local reactions and side‑effects immediately after the procedure and during the following follow-up visits: on the 3rd day, on the 7th day, at 3, 6, and 12 months, and at 2 and 3 years. The highest percentage of local reactions among registered cases immediately after the procedure was a bloated feeling (57.1%), pain (27.7%), symmetrical oedema (25.9%), reddening (21.4%), and bruising (16.1%). A total of 22.3% of patients were observed with one local reaction, 56.3% had two simultaneous reactions, and 11.6% experienced three local reactions.
Over 3 years, the following local reactions were observed among patients:
- Pain: 1.8% (periodically before menstrual cycle pain was slightly expressed and was felt for 1–2 days)
- Infection, inflammation: 2.7%
- Change to nipple sensitivity: 1.8%
- Induration: 1.8%.
No serious side-effects were observed immediately after the procedure, as well as over the 3 years. The most frequent side‑effects after the procedure were recorded as low‑grade fever (3.6%), weakness (3.6%), chill (0.9%), and headache (0.9 %). The 2.7% of patients who were examined with infection or inflammation during 3-year follow-up visit, were found to present as a result of a secondary reaction of the chronic inflammation process (e.g. purulent tooth inflammation, recurrent adnexitis in anamnesis). The conservative treatment with antibiotic therapy was considered effective in those cases. Sanitisation of the initial area was performed.
Efficacy assessments
Efficacy of the hydrophilic gel was evaluated using three assessment scales:
- Global Aesthetic Improvement Scale (GAIS) — assessed by both doctor and patient
- Psychological Comfort and Goal Achieving Scale (PCGAS): assessed by the patient only
- Evaluation of the effect duration (gel effectiveness): assessed by both doctor and patient.
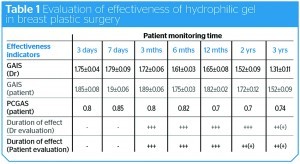
A 100% effect was observed at more than 2 years (doctor’s evaluation). Over 2–3 years, patients saw the need to perform an additional procedure in order to maintain the effect owing to physiological reasons (doctor’s GAIS=1.31±0.11; patient’s GAIS=1.52±0.09). The results are provided in the form of confidence intervals 3 years after the procedure. The evaluation of the effectiveness of hydrophilic gel in breast plastic surgery is presented in Table 1.
Conclusions
Hydrophilic gel (composed of 98% physiological sodium chloride solution and 2% polyamide) proved its efficacy and safety as a bodily dermal filler applied in breast plastic surgery. It is a good option both for doctor and patient, taking into consideration safety profile of the filler, its efficacy (long-lasting effect) and naturalness of breast soft tissues. No serious complications or allergies were observed during the follow-up period. No gel migration was reported.