Treatment
Subscision of each atrophic scar was performed 1 week prior to injection with CaHA to give a more uniform aesthetic effect. Radiesse was injected using a 27 gauge needle into the space left after subscision of the acne scar in the region of the mid- to deep dermis, although final placement also depended on the presence of fibrous and cystic tissue in this region of the skin. The total volume of CaHA used varied with each patient, with an end-point being agreed between patient and physician. It was decided not to record the amount of product used as this was not felt to be contributory to the final result. Histologic evaluation of cutaneous biopsies were not obtained before or during treatment, although it was offered to at least one of the patients who had a resultant adverse reaction. Betadine cleansing was used in most patients and doxycycline 100 mg for 2 days was given as prophylaxis in 11 patients, who were felt to be at risk of infection as they still seemed to have active acne. One patient, who had a previous photoallergic reaction to doxycycline, was prescribed Augmentin-Duo twice daily for 2 days.
Subjects were not excluded from the study on the basis of bleeding disorders or whether they were taking anticoagulants/anti-inflammatory agents, as it was felt that while the bruising may be unsightly, it would likely improve overall healing. Patients who had received synthetic collagen, HA, PMMA, CaHA, or autologous fibroblast injections to treated areas within the previous 6 months were excluded from the study. Clinical assessment scores were determined at each treatment session and follow-up visit. Patient satisfaction surveys and digital photography were used where they were appropriate to both parties, although both were considered subjective, with patients tending to focus on the smallest detail and physicians photographically favouring the better results. All patients were reviewed at 2 or 4 weeks post-treatment for a top-up of Radiesse, if required. It was noted that 17 patients required a top-up of at least 0.15 ml CaHA at one of the first two visits. Smaller amounts (< 0.1 ml) were not recorded, as it was felt that the patient may have seen some defects under deeper scrutiny that were initially missed during the procedure.
A series of 13 patients (7 male, 6 female; skin phototypes I–IV) with varying degrees of atrophic acne scarring were treated in a similar manner with low molecular weight cross-linked HA, and monitored over a 12-month period. Patients were randomly selected on the basis that they did not want to present for laser resurfacing as a treatment for their problem.
Results
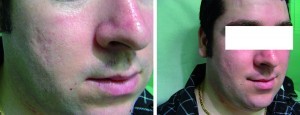
Figure 3 23-year-old male patient (left) before treatment with Radiesse, and (right) 2 weeks after treatment
Twenty-seven patients entered this 12-month study. Moderate to excellent clinical improvement was observed after 4 weeks in almost all of the patients studied (20 patients with score 4, 75–100%; six patients with score 3, 50–75%). One patient had score 1, 0–25% improvement, while no subjects scored either 0 or 5. At the 6-month assessment 12 patients (44% of total) had score 4 (75–100% improvement) and 11 patients (40%) had score 3 (50–75% ). At 12-month evaluation, six patients (22%) showed 75% improvement; 14 patients (48%) showed between 50% improvement, and five patients (18%) showed a 25% improvement in treated atrophic scars.
Thirteen patients entered the 12-month HA study. Moderate-to-excellent clinical improvement was observed after the initial treatments in almost all of the HA patients studied At 4-week assessment, nine patients still showed moderate-to-excellent results, but this had fallen off dramatically at 3-month assessment: only two patients had score 4 (75–100% improvement) and two patients (23% of total) had score 3 (50–75% improvement). At 6-month evaluation, 12 patients (92%) showed a 0–25% improvement.
The author felt both of the fillers initially provided a simple physical volumising effect. There was a longevity associated with the therapeutic effect of the calcium hydroxylapatite (CaHA), probably secondary to the duration of the filler and some level of neocollagenesis noted in other studies. There was little evidence of delayed biostimulatory effect of collagen formation owing to HA injections, although the physical act of injection and subcision was of some benefit to the patient.
Side-effects of treatment were mostly limited in the group to mild transient erythema, bruising or localised oedema. Some patients required top-up or remodelling on initial review. One 23-year-old male patient (skin type 4, of Asian origin) with minimally active acne on a previous trial developed cellulitis and later desquamation.
[pull_quote align=”right” ]CaHA is biosynthetically produced and does not elicit a chronic inflammatory or immune response.[/pull_quote]
Discussion
There are many methods that can be used in the treatment of atrophic acne scarring. Most tend to replace the volume lost by the atrophic effects of the acne. Newer injectable fillers are biocompatible and safer, and can provide an alternative means of treating acne scarring in patients not opting for laser resurfacing. The author has used the HA-based filler Matridex (BioPolymer GmbH & Co., Germany), CaHA, and the polyalkylimide Bio-Alcamid (Polymekon, Brindisi, Italy) for this purpose over the years. Some of these fillers simply provide a physical filling effect, while others induce a delayed collagen stimulatory effect (e.g. CaHA and poly-L-lactic acid). An ideal filling agent should restore atrophic volume and stimulate the dermis to synthesise new collagen for a long-lasting effect.
Based on the experience of this study, the author feels CaHA is a suitable product for this purpose, showing a clearly demonstrable benefit still present at 6–12 months. A comparative study performed with HA preparations showed the compound was not of medical or commercial benefit to either the physician or patient, with most of the product disappearing at only 6–12 weeks. More viscous forms of HA, such as HyaCorp (BioScience GmbH, Germany) showed no extra benefit.
Conclusions
CaHA is biosynthetically produced and does not elicit a chronic inflammatory or immune response. In vivo and in vitro studies have established the biocompatibility and safety of CaHA. No evidence of granuloma formation, ossification, or foreign body reactions have been found in long-term animal studies. CaHA implants have persisted intact at the injection site in areas such as the face at up to 12–18 months. No skin testing is required for the compound as company information states that no animal or animal products are used in the manufacture of the product, thus there is no risk of transmitting disease or causing allergic reactions in patients who are sensitive to common foods.
This study documents the efficacy of CaHA in the treatment of atrophic acne scars. The author is aware that these benefits may last 18 months or more.




