Charlene DeHaven discusses the skin repair and renewal properties of copper tripeptide-1 and its many applications in aesthetic medicine
In 1986, the NoBel Prize for Physiology or Medicine was awarded jointly to Stanley Cohen and Rita Levi-Montalcini for their discovery of the growth factors — nerve growth factor (NGF) and epidermal growth factor (EGF), respectively1. This sparked a new era of scientific expansion in all areas related to the regulation and direction of cell growth as mediated by growth factors and cytokines.
One of these techniques incorporated neonatal foreskins harvested during circumcision for the purpose of culturing epidermal cells8 for tissue intended for grafting. The use of the same basic technology, i.e. cell cultures obtained from neonatal foreskins, remains available today and has also been incorporated into cosmeceutical products9 through the use of cytokines and growth factors extruded into the nutrient media of such cell cultures. Other technologies use cytokines and growth factors from other types of discarded cultured human tissue, such as discarded fetal tissue10,11, and is also available in cosmeceutical products.
Not all of these cosmeceutical products share the same availability to consumers worldwide. European regulations are sometimes considered more stringent12.
Market interest in cosmeceuticals incorporating growth factors
Despite variances worldwide in the availability of products containing growth factors, market interest in this product category is high. Many cosmetic dermatologists are selling some type of growth factor skincare. Consumers seem better educated on product ingredients and are willing to spend money on products that they believe will deliver better results. ‘Consumers are particularly interested in product ingredients, including peptides and other growth factors that activate skin stem cells to boost collagen production and facilitate cell renewal’13.
Considering these factors, growth factor products with global cosmetic regulatory acceptance and positive ingredient effects well-documented in the medical literature, should have considerable consumer interest and marketability. Copper tripeptide-1 has been judged safe by the US Cosmetics Ingredient and Safety Review14 and is also available in the EU and worldwide.
History of copper tripeptide-1
Information about this compound was first published by Loren Pickart in 197515. In the many subsequent papers by Pickart and others, the compound was described as a growth factor with actions like hepatocyte growth factor (HGF)16. HGF is like other molecules in the family of growth factors in that it is a very long peptide chain with a large molecular weight and size. It is therefore more correct to describe copper tripeptide-1 as a growth factor analogue since it is composed of only three amino acids (a tripeptide). It is now known to fit into the family of cytokines. Because it is such an important messenger molecule with a wide range of properties and functions, it has sometimes been termed a ‘matrikine’17. However, in the literature it may still be referred to as a growth factor.
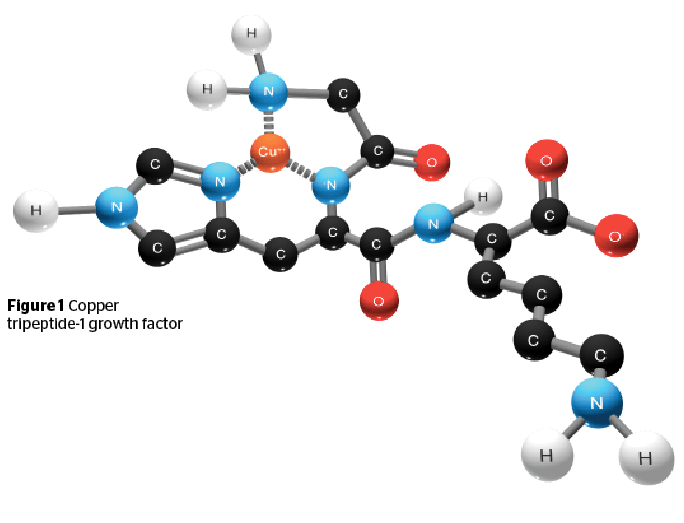
The molecular structure of copper tripeptide-1 is indicated by its name, i.e. a tripeptide chain consisting of glycine, histidine, and lysine (GHK) complexed with copper (Figure 1).
Natural role in the human body
As well as being present in wounded tissue, copper tripeptide-1 was also found to be a normal inhabitant of saliva, urine, and all collagenated tissue, as well as discovered to have important effects in maintaining and repairing all tissue types23.
Copper tripeptide-1 exists in forms both complexed with and without copper. It has a high affinity with copper, a metal associated with many enzymes in the human body that benefit tissue repair, inflammation, metabolism, and synthesis of vital molecular structures24. Copper metal ions themselves are also found in high concentrations around healing wounds and are therefore implicated in wound healing and inflammatory processes. Much of the benefit of copper tripeptide-1 relates to its ability to efficiently bind and transfer copper ions, a quality also related to its mitigation of oxidative stress25.
Skin penetration
The design of the skin serves as an effective physiochemical barrier. Penetration of long-chain peptides is problematic in spite of recent advances in drug delivery, which are not available for cosmeceuticals. In general, compounds greater than 500 Daltons in size are unable to penetrate the skin barrier26. Yet the majority of biologic processes are modulated by specific amino acid sequences. Short chain peptides with good barrier penetration that do not require advanced penetration enhancers or drug delivery systems are of great interest to the cosmetics industry27.
Actions and benefits for aesthetics
Copper tripeptide-1 may be considered one of the body’s emergency response molecules. It is released during injury and comes to the body’s aid when the processes below are activated.
Wound healing
Any tissue injury signals the release of copper tripeptide-1, which signals repair processes to begin. A huge body of scientific evidence supports the essential role of copper tripeptide-1 in the acceleration of wound healing in many human and animal wound types29-32, including surgical33,34, post-laser35, Mohs36, ischemic37, burns, large skin grafts38, hair transplants39, refractory venous stasis ulcers40, diabetic ulcers41, and others. Diabetic wounds healed three times faster in the presence of copper tripeptide-1 and time to re-epithelialization was shortened42,43.
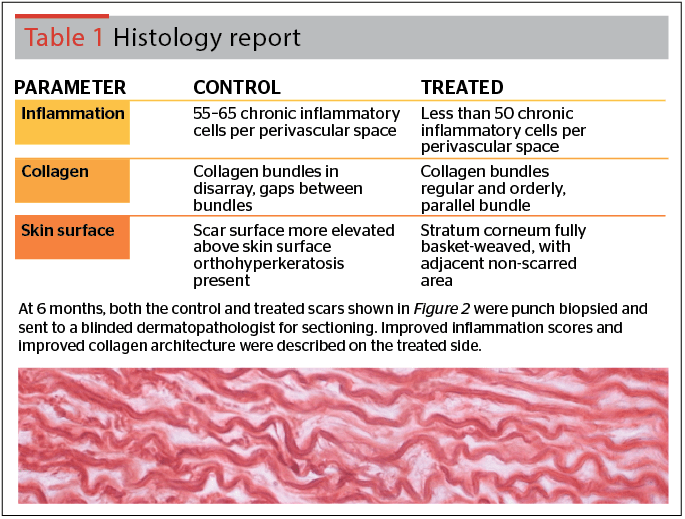
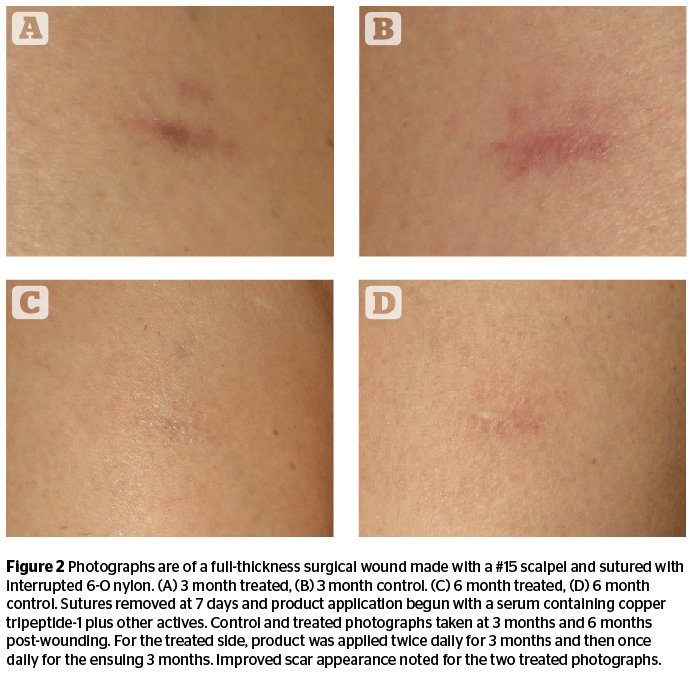
Figure 2 and Table 1 below illustrate the ability of copper tripeptide-1 to improve wound healing in skin for surgical wounds. The digital photographs shown in Figure 2 are of two identical full-thickness skin wounds made with a scalpel penetrating to the hypodermis. Both wounds were one centimeter in length. Sutures were removed at one week post-incision and application of a cosmeceutical product44 containing copper tripeptide-1 was initiated on the treated side. The photographs illustrate a much more cosmetically acceptable wound on the treated side at time periods of both 3 months and 6 months post-incision. These are compared to the control incision without product application that shows adequate wound healing but an inferior cosmetic result. These results substantiate the ability of cosmeceutical products containing copper tripeptide-1 to improve post-procedure scarring.
The incisional wounds of Figure 2 were then punch biopsied and sent to a fully blinded dermatopathologist for microscopy. The dermatopathologist’s description of the two wounds, both control and treated, are given in Table 1. With application of the cosmeceutical product containing copper tripeptide-1 to the treated incision, fewer inflammatory cells were seen and the stratum corneum exhibited a normal appearance. The stratum corneum was described as fully ‘basket-weaved’ and having the same appearance as the adjacent non-scarred area. Collagen bundles were regular and in orderly parallel bundles. These light microscopic findings further explain the improved cosmetic results seen with the treated scar in Figure 2.
Tissue remodelling and skin renewal
Furthermore, copper tripeptide-1 is active not only in wound healing but also for normal tissue remodelling. Studies have documented these regenerative effects in both human and animal studies. Animal studies have included rats, mice, pigs, rabbits, horses, dogs, and guinea pigs. The types of wounds studied in humans and animals have included gastrointestinal ulcers, liver injury from toxins, surgical wounds, burn wounds, ischemic wounds, bone fractures, punch biopsies, skin transplants, hair transplants, and tissue wounds evaluated in wound chambers. In skin, the specific effects of copper tripeptide-1 include increasing keratinocyte proliferation and normal collagen synthesis45,46, improving skin thickness47, skin elasticity and firmness48, improving wrinkles49, photodamage50 and glycation51, uneven pigmentation, skin clarity and tightening, and upregulating protective barrier proteins52,53.
Copper tripeptide-1 has decreased chemotherapeutic hair loss in both animals and humans and improved hair transplant survival and ‘take’. Furthermore, through effects on decorin54, new collagen made under the direction of copper tripeptide-1 assumes the correct anatomical configuration rather than a disorganized scar55. Matrix support structures of the dermis including collagen I, collagen III, and glycosaminoglycans56 increase as normal tissue configuration is restored in the presence of this growth factor. Thus it may be said that the properties of tissue remodelling and renewal associated with copper tripeptide-1 are ubiquitous throughout the human body and not unique to skin. The compound’s properties make it an attractive addition to cosmeceuticals both for anti-ageing effects and for optimizing post-procedure results.
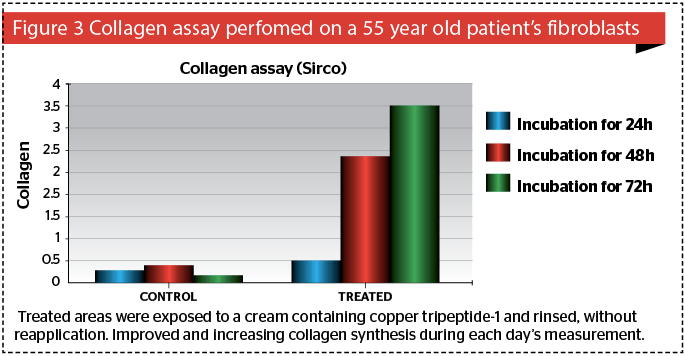
Likelihood of keloid formation is reduced and scar surface area is decreased59. Collagen production with this active was superior to creams containing vitamin C or retinoic acid60.
Anti-tumor effect and DNA repair
Copper tripeptide-1 has a strong anti-tumor effect61 on both primary tumor growth and metastases mediated by multiple possible mechanisms62 including decorin63,64, apoptosis65, and biologic chelation of the potentially pro-oxidant metals — iron66 and copper67,68. It protects DNA from the damaging effects of radiation during cancer treatments and photoaging. Radiated fibroblasts showed restoration of intact functioning after exposure to copper tripeptide-1. A variety of tumor cell lines have been shown to be suppressed or die in the presence of this compound while healthy cell lines continued to grow normally. This and other evidence implies that copper tripeptide-1 may be used in topical products to enhance normal tissue repair without fear of neoplastic transformation.
Stem cell anti-senescence
Survival of stem cells in the basal epidermis and other epithelial cells is promoted by tripeptide-169,70. This suggests it could have an important anti-ageing effect71,72 and, in fact, its anti-senescent effects73 have been demonstrated on a number of tissue types including skin74. Keratinocyte proliferation is encouraged through stem cell maintenance encouraged via copper tripeptide-175.
Antioxidant and anti-inflammatory effects
Many of the body’s intrinsic antioxidant systems, including superoxide dismutase, are induced by copper tripeptide-1 as demonstrated in healing wounds76.
Conclusion
Copper tripeptide-1 is a novel compound of great interest to aesthetics. This is related to its ubiquitous abilities not only in skin but also in other tissues to initiate tissue regeneration and wound repair for all types of wounds, increase collagen synthesis, effect DNA repair, quiet excess inflammatory processes, and act as an antioxidant. Its potential for improving post-procedure results and decreasing the velocity of ageing is obvious. Its safety profile is also quite remarkable and increases the level of interest. No adverse effects or issues have been found for copper-tripeptide-1 in spite of extensive research involving the compound since the early 1980s. As well as acting as a growth factor, it also has attractive anti-neoplastic effects that substantiate its safety profile for the ageing patient.






