Firas Al-Niaimi reviews the literature surrounding the use of botulinum toxin for a range of dermatological conditions, including hyperhidrosis, Darier’s disease, and Hailey-Hailey disease
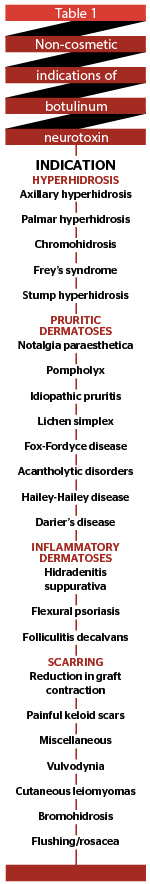
Botulinum neurotoxin (BoNT), colloquially referred to as ‘Botox’, is widely regarded as synonymous with the world of cosmetic dermatology. Since identification of Clostridium botulinum and the deadly neurotoxin it produces1, the ability of BoNT to paralyse muscles has been exploited for a host of cosmetic and non-cosmetic indications (Table 1). There is a growing list of indications for which BoNT has been used in dermatology beyond its common cosmetic indication.
Mechanism of action
There are seven structurally distinct isoforms of BoNT (A–G)2. BoNT-A (trade names: Botox, Dysport, Xeomin, Prosigne, Azzalure) is most commonly used, followed by BoNT-B (trade names: Myoblocs, NeuroBloc). BoNT-A and BoNT-B both consist of a protein comprising a heavy and light chain linked by a disulphide bond3. The heavy chain binds the neurotoxin to the cholinergic nerve terminal, while the light chain inhibits acetylcholine release4. BoNT-A and BoNT-B are pharmacologically distinguished by the type of protein the light chain cleaves, SNAP 25 in the case of BoNT-A and VAMP in the case of BoNT-B5.
BoNT’s most recognised mechanism of action is to inhibit acetylcholine release at the presynaptic neuromuscular junction. BoNT’s ability to prevent acetylcholine binding to postsynaptic receptors within the eccrine sweat gland and inhibit sweat production has led to its extensive use in the management of hyperhidrosis6. Furthermore, it has been suggested that BoNT exerts antinociceptive action via blocking the action of substance P, hence its more recent use in myofascial pain syndromes and headaches7.
 Hyperhidrosis
Hyperhidrosis
Hyperhidrosis, in which eccrine glands produce excessive secretions, can have a profound psychological impact upon quality of life. The excessive sweating is due to an abnormal physiological response to the hypothalamus, which can affect any eccrine sweat gland, but most commonly involving the palms, soles, and axillae8. The eccrine sweat glands in patients with hyperhidrosis are morphologically normal but are stimulated by excessive acetylcholine9.
A multi-centre, randomised placebo-controlled trial of BoNT-A treatment of bilateral primary axillary hyperhidrosis found that 94% of the treatment group (n=242) had greater than 50% reduction in sweating from baseline at weeks 1, 4, and 16 when treated with 50 U of BoNT-A (Botox, Allergan, Irvine, CA) compared with only 36% of the placebo group (n=78)10.
A multi-centre double-blind randomized controlled trial (RCT) evaluated the use of BoNT-A (versus placebo) in the treatment of primary axillary hyperhidrosis over 52 weeks (n=322) but here a variation in units was used (75 or 50 U), finding a 75% significant reduction in sweating (in the group given either 75 or 50 U) versus a 25% reduction in the placebo group. Interestingly, the duration of effect was found to be slightly longer in the 50 U group (205 days), lasting 8 days longer than the 75U group (197 days)11.
A randomized double-blind study of 11 patients evaluated the benefit of BoNT-A (120 U) in palmar hyperhidrosis, demonstrating a 26%, 26%, and 31% reduction in sweating at 4, 8, and 13 weeks, respectively (statistically significant), as measured by ninhydrin-stained sheet12. An intra-subject comparative study (n=19) evaluating the safety and efficacy of BoNT-A for palmar hyperhidrosis demonstrated statistically significant improvement with BoNT-A in one hand versus normal saline in the other13. With respect to side-effects following BoNT injection for palmar hyperhidrosis, a study of 427 patients found 275 (64%) reported local pain and 102 (24%) muscle weakness post BoNT-A injection (75–100U; Botox, Allergan)14.
Frey’s syndrome is a form of localized sweating of the parotid area associated with erythema29 during mastication, often attributed to trauma or surgery in that region15. The mechanism is thought to be secondary to the parasympathetic cholinergic nerve fibres aberrantly regenerating with sympathetic cholinergic receptors of the skin15. Treatment can be difficult, with options including atropine, antiperspirants, or preventative measures, such as neurectomy16. One study included seven patients with Frey’s syndrome (with at least 12 months of gustatory sweating) who were treated with 5–30 U BoNT-A (Botox, Allergan). Using a starch-iodine test to detect sweating, varying degrees of recurrence were noted. Some patients required no treatment at 12–24 months after a single dose of BoNT-A (12.5 U; Botox, Allergan) while others required repeated treatments over this time period15.
Stump hyperhidrosis, a condition frequently noted in leg amputee patients is often difficult to treat. Minimal results have been found with topical astringent agents and water iontophoresis. 100 U of BoNT-A (Botox, Allergan) injected into the stump was reported to prevent stump hyperhidrosis in a 44-year old amputee with diabetes. Initially, there was relapse at four months attributed to sweat transposition from higher up the stump. BoNT-A subsequently injected higher up the thigh improved the duration of anhidrosis (a total of 200 U given to the anterior and posterior superior thigh), with no relapse observed at seven months16.
A subsequent cohort of nine amputees reported a significant improvement in sweating at four weeks (p=0.027) and three months (p=0.020) following injection of BoNT-B 1750 U17.
Pruritic deratoses
A small double-blind, placebo-controlled study of 14 patients with pruritus induced by histamine prick test, showed that 5 U of BoNT-A (Botox, Allergan) significantly reduced pruritus intensity and affected area size in comparison to normal saline injections. Laser Doppler imaging carried out as part of the study also revealed that BoNT reduced the vasomotor action in the treated area25.
An intra-individual study of ten patients investigated the use of BoNT-A (mean dose 162 U; Botox, Allergan) for pomphoylx, using the untreated side as control. It found that 70% of patients subjectively reported an improvement with a 39% decrease in pruritus reported on the treated side versus a 52% increase on the untreated side. The authors hypothesized the increase in symptomatic pruritus could be due to compensatory hyperhidrosis. A further study involving six patients corroborated these findings with 100 U of BoNT-A (Botox, Allergan), suggesting that anhidrosis induced by BoNT can improve symptoms of pompholyx and reduce the number of relapses26. While both studies were limited by the modest sample size and subjective assessment tool, BoNT may be of use in recalcitrant cases of pompholyx.
An open pilot study of four patients investigating the effects of BoNT-A (20–80 U; Dysport, Ipsen Biopharmaceuticals Limited, Basking Ridge, NJ) on pruritus associated with lichen simplex found that pruritus not only subsided but some of the clinical lesions fully resolved within four weeks27.
Fox-Fordyce disease is a rare pruritic disorder most evident on skin with a high density of apocrine glands. Symptomatic pruritic papules have been shown to be responsive to 100 U of BoNT-A (Botox, Allergan) when injected into axillae, with an associated reduction in papules28.
Acantholytic disorders
Hailey-Hailey disease is an autosomal dominant acantholytic disorder concerning the mutation of the ATP2C1 gene, clinically manifesting as macerated flexural erythema29. Heat and sweat aggravate the disease, resulting in worsening of discomfort and pruritus symptoms. BoNT could rationally ameliorate all the above via its inhibition of acetylcholine and substance P. Several case reports have evidenced improvement of Hailey-Hailey with the use of BoNT-A (50–125U; Botox, Allergan)30–32. One case report compared BoNT-A with more conventional treatments of dermabrasion and Er:YAG laser therapy and found it to be equivalent, suggesting that BoNT may be a viable alternative to those not tolerating ablative treatments33.
Darier’s disease is another autosomal dominant acantholytic disorder, with mutation of the ATP2A2 gene. Manifestations include greasy warty papules in the flexures and heavily eccrine populated areas, which contribute to malodour. Evidence for BoNT use in Darier’s is limited with only a letter exampling a case of improvement in one patient who received an injection of 100 U BoNT-A (Botox, Allergan)34. The author has experience in the management of both Hailey-Hailey and Darier’s disease with BoNT injections.
Inflammatory dermatoses
Hidradenitis suppurativa (HS) is a chronic inflammatory dermatosis of the apocrine glands that typically affects the axillae and groin, while treatment is often challenging. There are several case reports showing improvement of symptoms with BoNT-A35–37, attributed to the inhibition of sweating. A 46-year old lady with chronic severe HS (Hurley stage 2) was reported to respond very well to axillary BoNT-A treatment (50 U) with a remission period of at least 12 months. This patient had previously been managed with antibiotics, the oral contraceptive pill, and surgical drainage of secondary abscesses. The authors hypothesized that BoNT-A inhibits apocrine secretions thereby preventing the inflammatory response evoked by follicular material from the pilosebaceous unit.
BoNT has been trialed in other inflammatory dermatoses that are thought to be exacerbated by excessive sweating. A pilot study of 15 patients with flexural psoriasis showed that 50–100 U of BoNT-A (Botox, Allergan) improved subjective symptoms and objective photographic evidence of the disease in 87% of patients at 2, 4, and 12 weeks38. Furthermore, patients with psoriasis are known to have a higher concentration of substance P receptors in their skin meaning BoNT should theoretically reduce pruritus by inhibiting substance P and hence prevent binding to the multiple receptors39.
Folliculitis decalvans is a primary neutrophilic cicatricial alopecia. One case study found favourable effects on hair regrowth post injection of 60–150 U of BoNT-A (Botox, Allergan). They hypothesized that BoNT-A inhibition of immune factors and cytokines reduced follicular inflammation40.
Scarring
BoNT can be used to reduce or soften troublesome scars, particularly those in areas with extensive muscular activity, such as the upper forehead. BoNT can be injected either at the time of scar formation (if anticipated) or at suture removal. Aside from muscular inhibition, BoNT is thought to inhibit the action of fibroblasts and hence reduce scar hypertrophy41. A randomized, double-blind, placebo-controlled study of primates found a significant improvement in scars injected with BoNT (when analyzed by plastic surgeons) in comparison to those that were not (p<0.01)42.
Keloid scars are a common complication of skin incisions, the incidence varying between races and also the location of the incision. BoNT is often quoted as an emerging treatment for keloid scars; however, there are few studies to support this. An 80-year old female with a painful keloid scar on the chest wall, previously unsuccessfully treated with triamcinolone, bupivacaine injections, lidocaine patches, gabapentin, and opioids, reported a significant improvement following injection of 100 U of BoNT-A (Botox, Allergan) into the scar43. The underlying mechanism of action remains largely unknown, with one study identifying no effect of BoNT upon cytokine and growth factor production of keloid tissue in cell culture44.
Miscellaneous indications
Several case studies have evaluated the role of BoNT injections in refractory cases of vulvodynia45,46 and found that BoNT-A is effective in blocking nociception associated with the vestibule. One case study was unable to assess the long-term effectiveness of BoNT as the patient opted for a vestibulectomy47. A separate study found that all subjects included (n=7) had a reduction in pain following injection of 20–40 U BoNT (Botox, Allergan), but there was variation in the number of injections required48. BoNT injection with progressive dilatation has been shown to be safe and effective in women suffering from vaginismus (10–150 U)49.

Lastly, there are several reports on the increasing use of BoNT in flushing and rosacea and this is an exciting approach to the treatment of these conditions52,53.
Discussion
BoNT has a multitude of non-cosmetic, dermatological indications for which it is able to either improve disease symptoms or treat the disease entirely. The use of BoNT in dermatological practice may be of particular value in cases of diseases refractory to other treatments.
Whilst there are many case examples within each disease category, there are few double-blinded randomised controlled trials, which are needed in order to compare BoNT to more commonly-used, established treatments. Trials involving larger numbers of patients, that include comparisons of dosage of therapy and subtypes of BoNT are lacking. Furthermore, the effect of the experience and training of the clinician administering BoNT on disease and symptom outcomes is yet to be explored.
The best evidence to date belies the use of BoNT in hyperhidrosis. Interestingly, some of the dermatological conditions we have discussed appear to respond to inhibition of sweating, such as Hailey-Hailey disease and stump hyperhidrosis in amputees, lending support to BoNT being used as an adjunct in recalcitrant disease. Other uses of BoNT may be seen in multi-disciplinary clinics, such as the use of BoNT for the treatment of anal fissures and retracted stomas that were causing peristomal dermatitis. Pruritus can be a result or an instigator of dermatological conditions. Evidence-to-date suggests BoNT can relieve pruritus, perhaps via its anhidrotic effects, therefore potentially easing the symptoms of many dermatological diseases. The cost of BoNT, transient nature of its response, and difficulty procuring funding from local commissioning groups will undoubtedly discourage its use by some dermatologists.
BoNT is a well-tested, relatively safe treatment and should be considered by dermatologists as a therapeutic possibility, particularly in recalcitrant cases of skin diseases.



 Hyperhidrosis
Hyperhidrosis