terimedix has designed and manufactured a new double-start fine-thread polycarbonate hub to allow us to produce a SILKANN aesthetic cannula that will fit onto an Allergan Syringe (non-standard thread) and perform under high injection forces.
The objective of this study is to determine an empirical value at which the cannula and syringe may become detached from each other.
The secondary objective is to analyse the performance and attachment properties of the fine thread hub to see whether it can also be used on a standard syringe fitting used by most other filler manufacturers.
Materials and methods
The tests were performed using a wide range of sodium hyaluronate/viscous products designed for use either in facial aesthetic procedures or ophthalmic surgical procedures:
-
1 Sodium hyaluronate 1%
-
2 Dermavisc cross-linked sodium hyaluronate 1 ml
-
3 Curamed CuraGel hydroxypropyl methylcellulose 2.4% 2 ml
-
4 Albomed hydroxypropyl methylcellulose 2% 2 ml
-
5 Allergan Juvéderm Ultra hyaluronic acid 1 ml
-
6 Sodium hyaluronate 1.6%
-
7 Microvisc Phaco sodium hyaluronate 0.85 ml
-
8 BBT Visco sodium hyaluronate 0.55 ml
-
9 Croma Neocrom sodium hyaluronate 1.4% 1 ml
-
10 Succeev Three hyaluronic acid 0.8 ml
-
11 Belotero Basic hyaluronic acid 1 ml.
Cannulae
-
1 27 G x 40 mm cannula with closed end and side port, with double-start fine-thread SILKANN polycarbonate hub — New Product
-
2 27 G x 40 mm cannula with closed end and side port, with double-start standard thread SILKANN polycarbonate hub — Existing Product
-
3 27 G x 40 mm cannula with closed end and side port, with standard luer lug polypropylene hub (equivalent to competitor products).
Equipment
Mecmesin MultiTest 2.5-xt pull/compression test machine; Emperor Force Testing System Software (V1.18-307 12/12/12).
Assumptions
For the purpose of this study, the maximum force that would be applied by a practitioner is 50 Newtons.
Methods
All cannulae are attached to the syringe finger-tight. The syringe is then mounted in the testing fixture and the test programme is run (programme shown above).
The programme is set up to replicate the use of a syringe by the user, so rather than a constant force being applied it is split up into three thrusts. The distance of the thrust is varied between syringes depending on the amount of product in the syringe. Each of the three thrusts is the same distance for the syringe being tested.
The load cell moves at a constant 500 mm/min for all testing and cannot be changed.
Once the programme is completed, the results are logged and the syringe and cannula
are inspected for sign of detachment, damage, or any other defects.
Results
Full results for each syringe and cannula combination are held on file at Sterimedix.
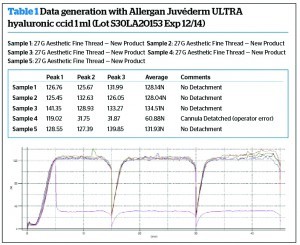
An example of the data generated is shown in Table 1.
Summary of results
The force required to detach a fine-thread hub (New Product) was not significantly different from the force required to detach a type 2 screw-thread hub (Exisiting Product) from the same syringe.
The lowest detachment force for the New Product was Succeev Three hyaluronic acid at a detachment of 93.06 N.
The lowest detachment force for the Existing Product was Curamed CuraGel hydroxypropyl methylcellulose 2.4% at a detachment of 117.63 N
The lowest detachment force because of a syringe collar detachment for the Exisiting Product was on Belotero Basic hyaluronic acid at 66.69 N. This did not occur on the New Product for the same or any other syringes.
The highest force achieved on the New Product was on Allergan Juvéderm Ultra hyaluronic acid at 134.51 N, but on a standard syringe is was on Microvisc Phaco sodium hyaluronate at 119.12 N.
The highest force achieved on the Existing Product was on a standard syringe Microvisc Phaco sodium hyaluronate at 122.47 N.
Discussion and conclusions
The International Standard for the minimum force required for detachment of the metal part of the cannula from the hub is 22 N.
The performance of the New Product (fine thread double-start polycarbonate hub) in conjunction with Allergan Juvéderm syringe (non-standard custom-thread) demonstrated that the integrity of the connection between the hub and syringe was acceptable. The performance of the New Product with all other syringes (standard thread) was also acceptable.
It is widely accepted that the desired injection force required for the injection of fillers should not exceed 25 N. It is therefore unlikely that the forces used in this test would be replicated during a procedure.
It can be concluded that the New Product’s unique properties may be used confidently with all manufacturers’ syringes, and is the only screw-thread polycarbonate hub compatible with all syringes currently available in the market.