Mukta Sachdev, MD, and Ninon Patrao, MD, review the current ingredients making waves in cosmeceuticals and particularly how they react with skin of colour
Ageing is an inescapable biological process, a natural physiological phenomenon, and the skin you see provides one of the first markers of the signs of ageing.
The three primary structural components of the skin dermis: collagen, elastin, and glycosaminoglycans, have been the focus of much of the research in this field. Preventing their degradation is necessary in order to mitigate the ageing process, i.e. the appearance of wrinkles1.
There are two main groups of agents that can be used as anti-ageing product components: antioxidants and cell regulators.
Antioxidants
Antioxidants form one of the most popular groups of cosmeceutical ingredients that are used today.Antioxidants reduce collagen degradation by decreasing the concentration of free radicals in tissues by blocking the oxidative processes in cells.
Vitamins C (L-ascorbic acid), B3 (Niacinamide), and E (α-tocopherol) are the most commonly used antioxidants due to their ability to penetrate the skin through their small molecular weight1.
Vitamin C in concentrations between 5 and 15% was proven to have a skin anti-ageing effect by inducing the production of collagen (Col-1 and Col-3), as well as enzymes necessary for the production of collagen, and inhibitors of matrix metalloproteinase-1 (MMP-1)2,3.
Niacinamide (vitamin B3) regulates cell metabolism and regeneration, and it is used in 5% concentration as an anti-ageing agent1. In certain studies, improvement of skin elasticity, erythema, and pigmentations after 3 months of topical treatment has been observed4,5.
Vitamin E (α-tocopherol) used as a component of skin products has anti-inflammatory and anti-proliferative effects in concentrations between 2 and 20%. It acts by smoothing the skin and increasing the ability of the stratum corneum to maintain its humidity, to accelerate the epithelialisation, and contribute to the photoprotection of the skin6.
The cell regulators act directly on the collagen metabolism and stimulate the production of collagen and elastic fibres. Cell regulators include vitamin A (retinol) and its derivatives (retinaldehyde, tretinoin) and peptides.
Vitamin A and its derivatives
Retinol is the naturally occurring form of vitamin A found in red, yellow, and orange fruits and vegetables. Retinol can be oxidised to retinaldehyde and then oxidised to retinoic acid, also known as tretinoin. It is this cutaneous conversion of retinol to retinoic acid that is responsible for the biological activity of some of the new stabilised vitamin A preparations designed to improve the appearance of photodamaged skin.
The conversion process consists of retinyl palmitate → retinol → retinaldehyde → retinoic acid (tretinoin).
The closer a compound is to retinoic acid on this cascade, the more readily it can be converted, and the more effective it becomes. Metabolism of retinaldehyde to retinoic acid occurs only by keratinocytes at a pertinent stage of differentiation, leading to a more controlled delivery of retinoic acid and weaker retinoid associated adverse effects like redness, dryness, and skin irritation. It has been shown to improve the feel of firmness and elasticity as well as reduce the appearance of fine lines, wrinkles, and rough skin8. Recent studies have revealed the efficacy and safety of new anti-ageing creams containing retinaldehyde at 0.1% and 0.05% that can be used to treat photoaged skin9.
Peptides
The natural ageing of skin results in decreased production and increased degradation of extracellular matrix proteins such as collagen, fibronectin, elastin, and laminin. Matrix metalloproteinases form an important family of metal-dependent endopeptidases that represent the primary class of enzymes responsible for the degradation of components of the extracellular matrix and thereby contribute to ageing10.
The extracellular matrix, in addition to providing structural support, also influences cellular behaviour such as differentiation and proliferation. These functions are mediated through small peptides (matrikines) derived from the proteolytic degradation of extracellular matrix proteins.
These polypeptides or oligopeptides are composed of amino acids and can imitate a peptide sequence of molecules, such as collagen or elastin. Through topical application, polypeptides have the ability to stimulate collagen synthesis and activate dermal metabolism11.
Cosmeceutical peptides may act as signal modulators of the extracellular matrix component, as structural peptides, carrier peptides, and neurotransmitter function modulators.
The advantages of using peptides as cosmeceuticals include their involvement in many physiological functions of the skin, their selectivity, and their lack of immunogenicity. However, they have a few drawbacks as well. The compounds are expensive and clinical evidence for efficacy is often weak. They also have poor absorption with high molecular weight and bind to other ingredients10.
Tranexamic acid
Tranexamic acid (TXA), an anti-fibrinolytic drug, is now gaining popularity as a depigmenting agent. It is a synthetic lysine amino acid derivative, which predominantly blocks the conversion of plasminogen to plasmin by inhibiting the plasminogen activator. This results in less free arachidonic acid production, and a reduction in the prostaglandin (PG) levels as well. Therefore, by reducing PG production, TXA reduces the melanocyte tyrosinase activity and plays an essential role in the treatment of melasma, ultraviolet-induced hyperpigmentation, and other post-inflammatory hyperpigmentation. It has been tried topically, orally, and intradermally in the management of melasma with minimal adverse effects. Orally, it is used in a dose of 250 mg twice daily for 3 months12.

The dermal and mixed variants of melasma are highly treatment-resistant. In such cases, TXA may be administered intradermally. The microneedling method proves to be efficacious in the intradermal delivery of the drug. In this method, multiple micro-injuries are made in the dermis, using a dermaroller and this facilitates the transport of substances through various transport channels, leaving the epidermis intact12.
Glutathione
Glutathione (GSH) was first discovered by Hopkins in 1921 in yeasts, and subsequently in other tissues. It is a tripeptide composed of L-cysteine, glycine, and glutamate that is synthesised intracellularly.
Proposed mechanisms of its action include inactivation of the melanogenic enzyme — tyrosinase, and influencing the switch from eumelanin to phaeomelanin. During melanogenesis, tyrosinase is responsible for the conversion of L-tyrosine to L-DOPA and subsequently to dopa-quinone, then the pathway bifurcates to produce eumelanin or phaeomelanin. At a critical point in the melanogenic pathway, thiols (cysteine and GSH) can react with L-dopaquinone to produce glutathionyl-dopa, or act as a reservoir of L-cysteine by conjugating with L-dopa-quinone to produce cysteinyldopa. These two thiol-dopa substrates serve as a precursor to enhance the switch from eumelanogenesis to phaeomelanogenesis, resulting in lighter skin pigmentation14. This effect of GSH on skin pigmentation was reported half a century ago, with darker human skin shown to exhibit lower levels of GSH than lighter skin. In addition, GSH can act to lighten the skin directly through the quenching of free radicals and peroxides that have been shown to induce tyrosinase activity. However, more evidence is needed to prove this unequivocally15.

Cysteamine
A derivative of the amino acid, L-cysteine, cysteamine hydrochloride, an older agent found to have depigmenting properties has been recently shown to have promising results. The malodour associated with it was initially a challenge but the development of agents to neutralise it while preserving its pigment suppressing properties has helped bring this agent to the forefront lately. The regimen involves a topical 5% formulation that is to be applied daily at night over the entire face and to be washed off after a 15 minute contact time. This is to be applied for 4 weeks followed by a maintenance regimen of twice weekly application for 4 more weeks. The use of cysteamine has been considered relatively safe in patients thus far although more studies are necessary to substantiate it.
Nanotechnology in cosmeceuticals
This is presently the most sought-after group of cosmeceuticals in the aesthetic field today from a commercial and marketable standpoint. The science and technology involved in it is used to develop or manipulate the particles in size range of 1 to 100 nm.
Nanomaterials are widely used in the preparation of antiwrinkle creams, moisturising creams, skin whitening creams, hair repairing shampoos, conditioners, and hair serums.
Novel nanocarriers such as liposomes, niosomes, nanoemulsions, microemulsion, solid lipid nanoparticles, nanostructured lipid carriers, and nanospheres have replaced the usage of the conventional delivery system. These carry the advantages of enhanced skin penetration, controlled and sustained drug release, higher stability, site-specific targeting, and high entrapment efficiency.
Despite their huge potential benefit, little is known about the short and long-term health effects from continued use. Researchers have indicated concern regarding the impact of increased use of nanoparticles in cosmeceuticals as nanoparticles can possibly penetrate through the skin and cause health hazards17.
Due to the production of a large number of oxygen species by nanoparticles, they may cause oxidation stress, inflammation, and damage to DNA. A number of ultrafine nanomaterials, such as carbon nanotubes, titanium dioxide, copper, and silver nanoparticles may be toxic to human tissues and cells. Titanium dioxide found in sunscreens has been demonstrated to cause damage to DNA, RNA, and fats within cells. There has been no stringent scrutiny imposed by the regulatory agencies for the approval and regulation of nanocosmeceuticals. Additionally, no clinical trials are required for the approval of nanocosmeceuticals, thus raising a concern of toxicity after use18.
Nutraceutical and nutricosmetics
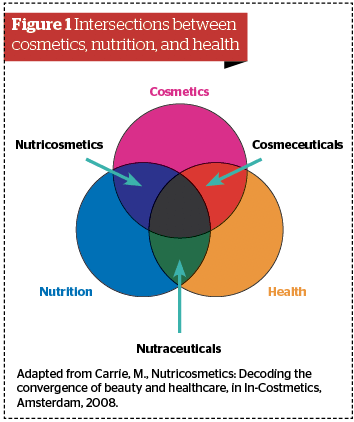
Cosmeceuticals are at the intersection between cosmetics (products that simply clean and beautify) and health care (products that cure and heal) and are defined as topical cosmetic products that claim to have medical- or pharmaceutical-like benefits. Their examples include anti-ageing products, skin whiteners or brighteners, acne aids, whitening toothpastes, anticellulite products, or antiperspirant. Alternatively, nutriceuticals are at the intersection between nutrition and health care and are foods or beverages providing health benefits including the prevention or treatment of disease (also called ‘functional foods’)19.
The term nutraceuticals was coined from ‘nutrition’ and ‘pharmaceutical’ by Stephen Defelice MD, in 1989. The functional food concept can be defined as food products to be taken as part of the usual diet in order to have beneficial effects that go beyond basic nutritional function20.
Nutricosmetics, on the other hand, can be defined as ingestible products (pills or capsules, tablets, liquids, granulates, or foods) that are formulated and marketed specifically for beauty purposes. Such products are at the intersection between nutrition and personal care, and they should not be confused with cosmeceuticals or nutraceuticals19.
Nutraceuticals became a major focus of food and pharmaceutical companies in the 1990s. Examples of such oral drug-like products are cholesterol-lowering, diabetes management, tartar control, digestive aid, and energising and fitness-enhancing products.
Finally, nutricosmetics emerged as a segment of nutraceuticals, which first gained popularity in Japan and Europe. The development and market are dominated by beauty firms, with only a few pharma or food entrants19.
The categories defined as nutraceuticals include:
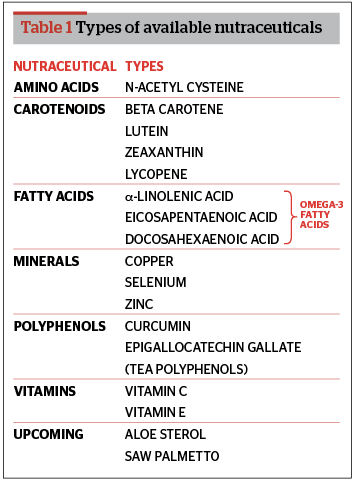
- Nutrients: substances with established nutritional functions, such as vitamins, minerals, amino acids, and fatty acids
- Herbal: botanical products as concentrates or extracts
- Dietary supplements: reagents derived from other sources (e.g., pyruvate, chondroitin sulfate, steroid hormone precursors) serving specific functions, such as sports nutrition, weight-loss supplements, conventional fortified foods, and meal replacements19.
Nutricosmetics are usually based on combinations of the following ingredients: carotenoids, polyphenols, several vitamins, soy extracts (e.g., polyphenolic isoflavones), micronutrients, glycopolyglycans, amino acids, other plant-based elements (e.g., herbs), and polyunsaturated fatty acids (e.g., fish oils). The top nutricosmetic ingredients are collagen, co-Q10, grape-seed extract, green tea, lutein, lycopene, marine complex (deep-sea fish ingredient), omega-3, superfruits (e.g., acai), vitamins A, C, and E, and zinc19.

Keeping in mind the relentless and sometimes gratuitous aesthetic market today and the breakneck rate of technological advancement, it is essential that patients are counselled about the nature and availability of cosmeceuticals and authentic, medically certified supplements, and their prudent use if and when the need arises.
Conclusions
The impact of social media and marketing today for beauty and skin care in all skin types is profound. In patients with skin of colour, the obsession with skin fairness and lightening remains a challenge for skin care professionals.
It is imperative that aesthetic doctors initiate a language revolution while treating such patients with special emphasis given to the use of terminologies such as even tone, radiance, and rejuvenation, and also to find a healthy balance between patient expectations and what can be reasonably achieved with professional treatment.
Declaration of interest None
Figure 1 Adapted from Carrie, M., Nutricosmetics: Decoding the convergence of beauty and healthcare, in In-Costmetics, Amsterdam, 2008.)
References:
- Ganceviciene R, Liakou AI, Theodoridis A, Makrantonaki E, Zouboulis CC. Skin anti-aging strategies. Dermatoendocrinol. 2012 Jul 1;4(3):308-19
- Nusgens BV, Humbert P, Rougier A, Colige AC,Haftek M, Lambert CA, et al. Topically applied vitaminC enhances the mRNA level of collagens I and III,their processing enzymes and tissue inhibitor of matrixmetalloproteinase 1 in the human dermis. J Invest Dermatol 2001; 116:853-9
- Kockaert M, Neumann M. Systemic and topical drugs for aging skin. J Drugs Dermatol 2003; 2:435-41
- Bissett DL, Miyamoto K, Sun P, Li J, Berge CA. Topical niacinamide reduces yellowing, wrinkling, red blotchiness, and hyperpigmented spots in aging facial skin.Int J Cosmet Sci 2004; 26:231-8
- Kerscher M, Buntrock H. Anti-aging creams.What really helps?. Hautarzt 2011; 62:607-13
- Zhai H, Behnam S, Villarama CD, Arens-Corell M, Choi MJ, Maibach HI. Evaluation of the antioxidant capacity and preventive effects of a topical emulsion and its vehicle control on the skin response to UV exposure. Skin Pharmacol Physiol2005; 18:288-93
- Sander, C.S., Chang, H., Salzmann, S., Muller et al Photoaging is associated with protein oxidation in human skin in vivo. J. Invest. Dermatol 2002;118:618–625
- Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G.Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1(4):327-48
- Kwon HS, Lee JH, Kim GM, Bae JM. Efficacy and safety of retinaldehyde 0.1% and 0.05% creams used to treat photoaged skin: A randomized double-blind controlled trial. J Cosmet Dermatol. 2018 Jun;17(3):471-476
- Pai VV, Bhandari P, Shukla P. Topical peptides as cosmeceuticals. Indian J Dermatol Venereol Leprol. 2017 Jan-Feb;83(1):9-18
- Lupo MP, Cole AL. Cosmeceutical peptides. Dermatol Ther 2007; 20:343-9
- George A. Tranexamic acid: An emerging depigmenting agent. Pigment Int 2016;3:66-71
- Ebrahimi B, Naeini FF. Topical tranexamic acid as a promising treatment for melasma. J Res Med Sci 2014;19:753 7
- Davids LM, Van Wyk JC, Khumalo NP. Intravenous glutathione for skin lightening: Inadequate safety data. S Afr Med J. 2016 Jul 6;106(8):782-6
- Halprin KM, Ohkawara A. Glutathione and human pigmentation. Arch Dermatol 1966;94(3):355-357.
- Watanabe F, Hashizume E, Chan GP, Kamimura A. Skin-whitening and skin-condition-improving effects of topical oxidized glutathione: A double-blind and placebo-controlled clinical trial in healthy women. Clin Cosmet Investig Dermatol 2014;7:267
- Kasraee B, Mansouri P, Farshi S.Significant therapeutic response to cysteamine cream in a melasma patient resistant to Kligman’s formula. J Cosmet Dermatol. 2019;18:293–295
- Kaul S, Gulati N, Verma D, Mukherjee S, Nagaich U. Role of Nanotechnology in Cosmeceuticals: A Review of Recent Advances. J Pharm (Cairo).2018
- E. Starzyk, A. Frydrych, and A. Solyga, ‘Nanotechnology: does it have a future in cosmetics?’ SO¨FWJ ournal. 2008vol;134(6):42–52
- Taeymans, Clarys, O. Barel.Use of Food Supplements as Nutricosmetics in Health and Fitness – A Review. Handbook of Cosmetic Science and Technology, Edition: 5th, Chapter: 5 2014;583 – 596
- Chauhan B, Kumar G, Kalam N, Ansari SH. Current concepts and prospects of herbal nutraceutical: a review. J Adv Pharm Technol Res. 2013;4:4–8
- Souyoul SA, Saussy KP, Lupo MP. Nutraceuticals: A Review. Dermatol Ther (Heidelb). 2018;8(1):5-16








