ABSTRACT
Submental fat, which may occur as a result of genetics, lifestyle or ageing, can cause an undesirable chin profile. Invasive methods may not be suitable for all patients wishing to reduce unwanted submental fat, but a clinically validated non-surgical alternative is lacking. ATX-101, a proprietary, injectable formulation of synthetically derived deoxycholic acid, has been investigated for the reduction of submental fat. ATX-101 is the subject of a rigorous clinical development programme to evaluate its efficacy and safety for the reduction of unwanted submental fat. Phase I studies established the mechanism of action and drug profile of ATX-101 in healthy volunteers. When injected into subcutaneous fat tissue, ATX-101 caused localised adipocytolysis (the destruction of fat cells) and led to the removal of cellular debris and lipids without drug accumulation. In phase II studies, clinicians reported a significantly greater reduction in submental fat with ATX-101 compared with placebo (p<0.05), and improved patient satisfaction with the appearance of the face and chin after treatment (p<0.05). In two European phase III studies that together included more than 700 adults with moderate or severe submental fat, a clinically and statistically significant reduction in clinician-rated submental fat was evident with ATX‑101 compared with placebo (p<0.001), and significantly more patients were satisfied with the appearance of their face and chin after treatment (p<0.001). Patients reported feeling happier, less bothered and less self-conscious about their submental fat with ATX-101 compared with placebo, and felt that they looked less old and less overweight after treatment. ATX‑101 was well tolerated. The most common treatment-related adverse events occurred at the injection site, were predominantly transient, and generally resolved during the interval between treatments. In the future, ATX-101 may provide a non-surgical option for the reduction of submental fat.
The presence of subcutaneous fat beneath the chin, known as submental fat, may result in an undesirable chin profile and loss of mandibular line definition1,2, giving rise to what is commonly referred to as ‘double chin’. This unwanted submental fat may develop owing to factors related to genetics, lifestyle or the ageing process1,3. Neck-lift and liposuction of the neck and chin are established means of addressing unwanted submental fat1. However, surgical approaches may lead to postoperative complications or long recovery times and might, therefore, not be suitable for all patients. Although injectable fat-reducing formulations and energy devices are available4, none has a sufficient level of clinical evidence to support its use, leaving plastic surgeons and dermatologists without a robust, non‑surgical option to offer their patients. ATX-101, currently under development by KYTHERA Biopharmaceuticals (Westlake Village, CA), may fill this gap.
ATX-101 and its mode of action
ATX-101 is an injectable adipocytolytic therapy that causes the destruction of fat cells. ATX-101 is a proprietary, injectable formulation of synthetically derived deoxycholic acid (DCA). DCA interacts with cell membranes, leading to cellular signalling responses5,6, membranolysis7 and, ultimately, cell death8. Importantly, when given at appropriate concentrations, the activity of DCA is attenuated in cells that are rich in albumin, such as skin and muscle9, owing to its high affinity for protein binding10. Consequently, adipocytes, which have a low protein:lipid ratio, are more susceptible to DCA than other surrounding tissues9.
Adipocytolysis induces a local inflammatory response in the treatment area9,11,12. Infiltrating macrophages remove the cellular debris, including lipids, over a period of approximately 4 weeks13. In healthy volunteers, plasma lipid levels showed a transient increase after ATX-101 injection, similar to that observed after a meal, but without any significant changes in the overall serum levels of total cholesterol, total triglycerides or free fatty acids14. DCA does not accumulate in the treatment area because it is rapidly eliminated into the enterohepatic circulation to join the endogenous bile acid pool9. In a phase I study in healthy volunteers, ATX-101 demonstrated predictable pharmacokinetic properties without evidence of accumulation in the systemic circulation15.
Desired clinical standard in submental fat reduction
Unlike other unapproved injectable therapies for localised fat reduction, ATX-101 is the subject of an evidence-based clinical development programme, conducted in accordance with rigorous international standards, to evaluate its efficacy and safety for the reduction of unwanted submental fat. Proof-of-principle was established through a series of preclinical experiments and phase I and II studies, which laid the foundations for randomised, placebo-controlled phase III trials in patients.
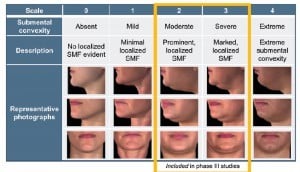
Outcomes in phase II and III studies were evaluated primarily by means of carefully developed rating scales for clinicians and patients that were designed with the goals of therapy in mind. A clinician-reported scale to describe the extent and severity of submental fat — the Clinician-Reported Submental Fat Rating Scale (CR‑SMFRS) (Figure 1) — was developed. Importantly, patients were also asked to rate their satisfaction with the appearance of their face and chin before and after treatment (Subject Self-Rating Scale — SSRS) as a co‑primary endpoint alongside the CR-SMFRS. Additional patient assessments of the treatment effect were recorded via the Patient-Reported Submental Fat Rating Scale (PR-SMFRS), through which the amount of unwanted chin fat was rated; the Patient-Reported Submental Fat Impact Scale (PR-SMFIS), which considered the psychological impact of unwanted submental fat and the effect of treatment; as well as a series of other patient-focused questions. Changes in submental fat were quantified using magnetic resonance imaging (MRI), and calliper measurements of submental fat thickness were also recorded as supportive measures of efficacy. Clinicians also rated skin laxity using the Skin Laxity Rating Scale (SLRS). Together with thorough evaluations of safety throughout each clinical trial, these measures were used with the aim of establishing the clinical profile of ATX-101 for submental fat reduction.
Performance in phase II studies
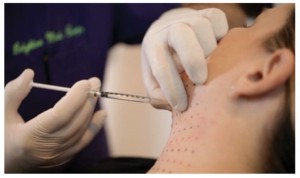
Two international, multicentre, randomised, double-blind, placebo-controlled phase IIa (NCT00618722 and NCT00618618)16 studies and one phase IIb (NCT01032889)17 study were conducted to evaluate the efficacy and safety of ATX–101 for the reduction of unwanted submental fat. Adult patients were included if they had prominent or marked submental fat that they considered undesirable and had no prior history of interventions for its reduction. ATX-101 doses of 1, 2 and 4 mg/cm2 or placebo were administered via a series of injections, spaced using a grid (Figure 2), into the submental area at a maximum of four (phase IIa) or six (phase IIb) treatment sessions each separated by 4 weeks16,17.
In the phase IIa studies, efficacy was primarily evaluated by clinicians using the CR-SMFRS and by patients via the SSRS. In a pooled analysis, clinicians reported a significantly greater reduction in submental fat compared with placebo with all ATX-101 doses at 4 and 12 weeks after the final treatment (p<0.05)16. Submental fat reduction with ATX-101 was not associated with worsening of skin laxity (SLRS) in the majority of patients. Patient satisfaction with the appearance of the face and chin after treatment (SSRS) was also significantly higher with ATX-101 than the placebo group 4 weeks after the final treatment (p<0.05)16. In a long-term follow-up analysis of patients enrolled in these trials, >90% had a sustained reduction in submental fat, and >80% had the same or improved satisfaction with their face and chin appearance at >2 years after treatment compared with the end of the original studies18.
In the phase IIb study, efficacy was evaluated using CR–SMFRS and SSRS scores, as well as the PR–SMFRS and PR-SMFIS scores17. Objective assessment was provided by MRI scans of the submental thickness and volume before and after treatment. Patients receiving ATX–101 2 mg/cm2 had significantly greater reductions in submental fat than those receiving placebo from week 4 onwards, as assessed by clinicians (CR-SMFRS; p<0.05). Twelve weeks after the final treatment, significantly greater reductions in the amount of submental fat were also reported by patients (PR-SMFRS; p<0.001 vs placebo), and this was supported by reductions in submental fat thickness and volume observed using MRI (p=0.006 vs placebo). Patients who received ATX‑101 felt significant satisfaction with the appearance of their face and chin (SSRS; p<0.001 vs placebo) and reported significant improvements in the psychological impact of their submental fat compared with patients who received placebo (PR-SMFIS; p<0.001 vs placebo)17.
In these studies, ATX-101 was well tolerated16,17. Overall, adverse events associated with ATX-101 treatment were localised to the treatment area (e.g., 90.7% in the phase IIb study). The most commonly reported events were injection-site pain, bruising, anaesthesia, induration, swelling, erythema, oedema, pruritus and paraesthesia16,17. Most adverse events were mild or moderate in intensity (e.g., 88.4% in the phase IIb study) and bore a temporal relationship to dose administration. The higher dose provoked more adverse events that were perceived as severe by a small proportion of patients, but all adverse events resolved within the 4-week treatment interval, and no treatment-related systemic adverse events occurred. In the 2-year follow‑up study, no new clinically significant treatment-related adverse events were reported18.
Randomised phase III clinical studies
Significant improvements in submental fat and patient satisfaction
The phase II results indicated that ATX-101 doses of 1 mg/cm2 and 2 mg/cm2 were likely to offer the best balance of efficacy and tolerability, and these doses were tested in two European, randomised, placebo‑controlled phase III clinical studies (NCT01305577 and NCT01294644) that together enrolled more than 700 patients19,20. Patients were eligible to participate if they were aged 18–65 years and had moderate or severe submental fat (grade 2 or 3 out of a maximum of 4 on the CR-SMFRS), and expressed dissatisfaction with the appearance of their face and chin (SSRS score 0–3). The main exclusion criteria encompassed any previous intervention to treat submental fat and anatomical features or previous trauma liable to interfere with efficacy evaluation or cause an unacceptable aesthetic outcome. After screening and baseline evaluations, patients received up to four treatment sessions spaced 28 days apart — this interval was largely sufficient for resolution of any local tissue response, based on results of a phase I histology study13.
At each treatment session, a maximum volume of 10 ml of study drug (0.2 ml per injection) was administered, with the number of injections depending upon the size and distribution of each patient’s submental fat. Injections were spaced 1 cm apart using a grid to provide an appropriate distribution across the submental area. The primary efficacy outcomes, assessed against baseline at 12 weeks after the final treatment, were a ≥1-point reduction in the 5-point clinician rating scale (CR-SMFRS) and a final score of at least 4 on the 7-point SSRS, indicating patient satisfaction with the appearance of their face and chin. Calliper measurements of the submental area and patient‑reported outcome measures, such as PR‑SMFRS, PR‑-SMFIS and other patient self-rating and quality-of-life questions, were also used. Safety (adverse events, clinical laboratory tests, vital signs, concomitant medications and physical examinations) was monitored throughout the study.
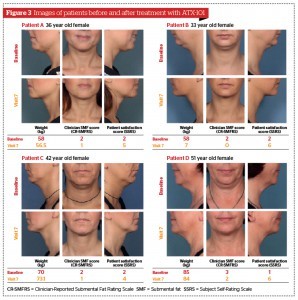
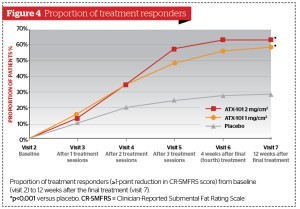
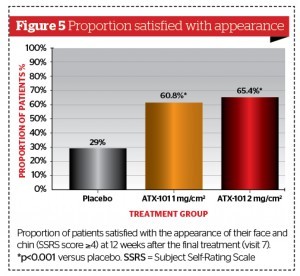
In both studies, a clinically meaningful and statistically significant reduction in clinician-rated submental fat was evident with both ATX-101 doses compared with placebo; between 58% and 65% of ATX‑101 patients had a ≥1 point improvement in CR‑SMFRS, compared with 23–35% with placebo (p<0.001)19,20. In addition, significantly more ATX-101 recipients than those who received placebo were satisfied with the appearance of their face and chin after treatment (SSRS score ≥4: 53–68% vs 29%; p<0.001). There was a significant reduction with ATX-101 versus placebo in the thickness of submental fat measured by calliper (p<0.001)19,20. Representative photographs of patients treated with ATX-101 are displayed in Figure 3. In a pooled analysis of the data from both studies, significantly greater proportions of patients had improvements in CR-SMFRS (59% and 64% with ATX-101 1 mg/cm2 and 2 mg/cm2, respectively, vs 29% with placebo; p<0.001; Figure 4) and SSRS (61% and 65% with ATX-101 1 mg/cm2 and 2 mg/cm2, respectively, vs 29% with placebo; p<0.001; Figure 5)21.
The efficacy of ATX-101 was also supported by secondary patient-reported outcomes in both studies. Consistent with the clinicians’ evaluation of submental fat reduction, patients receiving ATX-101 also reported a significant improvement in PR-SMFRS score compared with those who received placebo (65–74% vs 32–44%; p<0.001). Patients in both ATX-101 groups also felt a greater improvement in the psychological impact of treatment (PR-SMFIS) compared with the placebo group; they felt happier, less bothered, and less self-conscious about their submental fat after receiving ATX-101, and felt that they looked less old and less overweight after treatment19,20. As a result, a significantly higher proportion of patients who received ATX-101 were extremely satisfied with the treatment they received compared with those who received placebo (p=0.049 for the 2 mg/cm2 dose)19,20.
Adverse events
ATX-101 was well tolerated. The most common treatment-related adverse events occurred at the injection site (93–99%) and included pain, swelling, oedema, erythema, bruising, numbness and induration. These events are expected given the mechanism of action of the drug and the nature of the injections19,20, and occurred with a higher incidence in both ATX-101 treatment groups compared with the placebo group (91–99% vs 51–79%). Nevertheless, they were predominantly transient in nature and generally resolved within the 4-week interval between treatments. Most adverse events were mild to moderate in intensity, with the exception of injection-site pain, which was mostly moderate to severe but had a median duration of only 1 day. No adverse events with a probable relationship to ATX-101 occurred outside of the treatment area. No relevant trends in clinical and laboratory safety parameters occurred.
ATX-101 in clinical practice
The ATX-101 clinical development programme is building clinically meaningful and consistent evidence to enable non-surgical and durable reductions in submental fat that translate into improved patient satisfaction. Surgical procedures and liposuction will remain important options for submental fat reduction, particularly when other anatomical features of the chin and neck are to be addressed in tandem. However, a minimally invasive alternative for patients who are at high risk of developing post-surgical complications or may face extensive recovery periods would be an attractive addition to the aesthetic physician’s toolkit. It remains to be seen which types of patient may benefit most from ATX-101 treatment, and which may be more appropriate for other modalities. Pooled analyses of the two European studies, additional results from two large phase III trials based in the United States, as well as a long-term follow-up of these patients, will provide more information on this and other questions.
It should be noted that ATX-101 is currently an investigational pharmaceutical product and is not licensed for medical use. ATX-101 is being tested exclusively for the reduction of submental fat and not in other body areas. This is because adipocytolytic therapy remains an investigational approach at this stage. However, based on the results of clinical studies to date, ATX-101 may in the future offer an evidence-based alternative to invasive methods of submental fat reduction.